Aluminum is famously easy to overheat and deceptively hard to master. One moment it looks solid, the next it slumps, warps, or the weld collapses at the edge. If you work in a small shop or garage, understanding the melting point of aluminum—and how alloys actually soften and melt over a range—can save parts, time, and fingertips.
Here’s the deal: pure aluminum has a single melting point, but most shop materials aren’t pure. They’re alloys, and alloys transition through a “mushy” semi-solid zone between two temperatures. That range is exactly where many welding and brazing problems begin.
Key takeaways
- Pure aluminum melts at 660.32 °C (1220.58 °F), per the authoritative NIST Chemistry WebBook; most shop alloys do not melt at a single point.
- Alloys have a solidus and a liquidus. Between them is a semi-solid “mushy zone” where support drops quickly—critical for welding, brazing, and casting control.
- For welding and fabrication, the heat-affected zone (HAZ) in common 6xxx alloys often softens before you ever see a puddle. Managing heat input and travel speed is essential.
- Many aluminum alloys start losing meaningful strength above roughly 150–200 °C (300–392 °F). Treat service temperatures in this range with caution and check datasheets.
- Practical takeaway: rely on alloy-specific data, control heat diligently, and fixture to resist distortion; avoid universal “one-temperature-fits-all” rules.
What melting point really means in aluminum
For elemental aluminum, the melting point is well established: 660.32 °C (1220.58 °F). That value comes from the US National Institute of Standards and Technology, see the aluminum entry in the authoritative NIST Chemistry WebBook (Tfus ≈ 933.45 K).
But shop aluminum is typically an alloy. Alloys don’t jump from fully solid to fully liquid at a single temperature. Instead, they begin to melt at the solidus and finish melting at the liquidus. In between sits the semi-solid or “mushy” zone. Think of it like wet sand: it can look intact yet lose load-bearing capacity with just a bit more heat.
Below is a simplified aluminum–silicon phase diagram to visualize those ideas. Note the eutectic point near 12.6% Si at about 577 °C, and a hypoeutectic composition around 7% Si (similar to A356) with a clear mushy range bounded by solidus and liquidus. For reference on eutectic behavior in the Al–Si system, see the overview in ASM’s Alloy Phase Diagrams content on eutectic systems (ASM International).
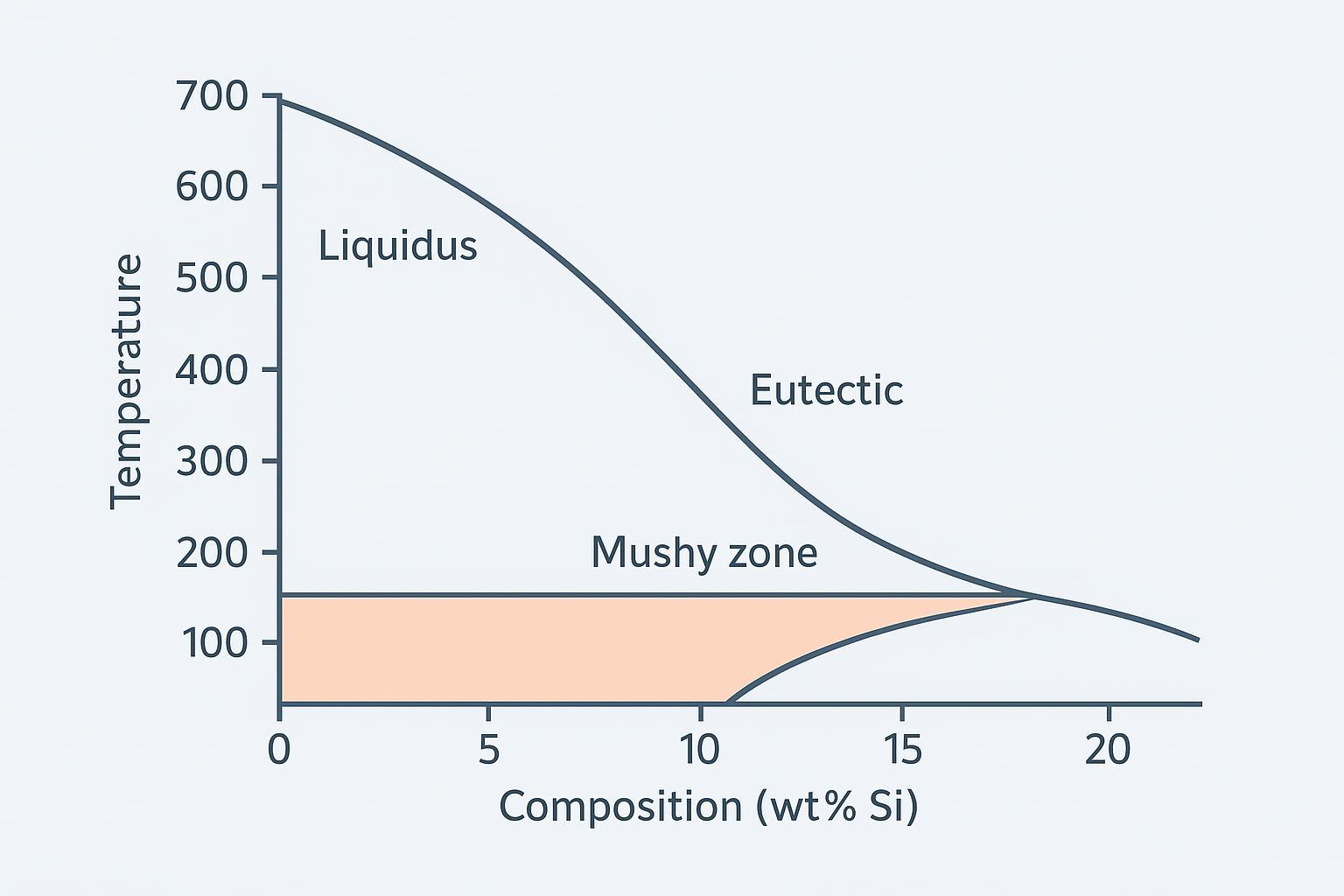
Quick-reference alloy ranges and notes
The table below lists common shop alloys with typical solidus and liquidus temperatures. Values vary by exact composition and source; always confirm with the latest datasheet or handbook.
| Alloy | Solidus °C (°F) | Liquidus °C (°F) | Shop notes |
|---|---|---|---|
| 5052-O (Al–Mg) | ~607 (1125) | ~649 (1200) | Readily weldable; watch for HAZ softening if over-heated. Data: ASM/MatWeb 5052-O. |
| 6061 (heat-treatable 6xxx) | ~582 (1080) | ~652 (1205) | Strong in T6 but prone to HAZ softening/overaging. Data: ASM/MatWeb 6061-T6. |
| A356.0 (Al–Si–Mg casting) | ~555 (1035) | ~615 (1135) | Hypoeutectic; clear mushy range affects feeding and weld edges. Data: ASM Handbook A356. |
Why include both °C and °F? Because small errors in conversion propagate during setup. Dual units help avoid misreads and speed shop decisions.
Why the melting range matters for welding and the HAZ
Aluminum conducts heat fast and melts relatively low, so temperatures near the solidus creep up quickly beyond the arc’s footprint. In heat-treatable alloys like 6061-T6, the heat-affected zone can lose strength significantly due to overaging or localized annealing, even when the bead itself looks fine. Many training notes point out that tensile tests often fail in the HAZ for poorly controlled procedures. For an accessible discussion of HAZ softening and procedure pitfalls, see this ESAB technical explainer on aluminum HAZ behavior.
Practical controls you can apply today:
- Control total heat input. Favor stringer beads and higher travel speeds over wide weaves. Consider pulsed MIG/TIG modes to reduce average heat.
- Keep preheat conservative on heat-treatable alloys and mind interpass temperature. Typical shop practice for 6xxx aims to minimize preheat and keep interpass temperatures modest; always qualify per code and datasheet.
- Fixture smartly. Aluminum’s high conductivity and expansion encourage distortion. Use clamps, strongbacks, and balanced welding sequences to limit movement.
- Prepare joints meticulously. Cleanliness and fit-up reduce arc time and rework—less time near the solidus means a healthier HAZ.
Below is a simple diagram showing weld metal, HAZ, and base metal in an aluminum plate, with the HAZ band highlighted where softening is most likely in heat-treatable alloys.
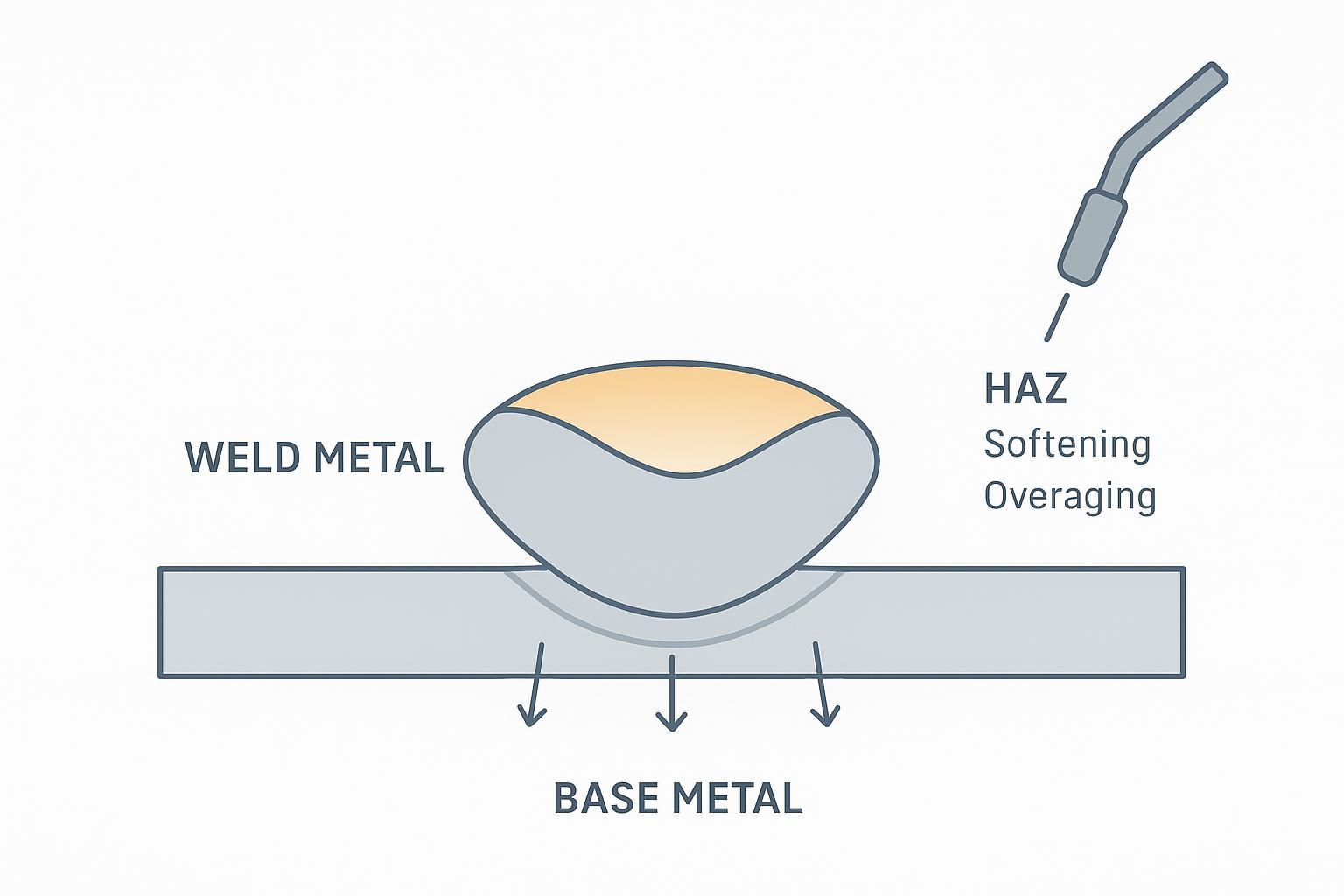
For broader background on preheat, interpass, and post-weld treatments, see ESAB’s training notes on preheating and post-weld heat treatments, and their overview of code context around aluminum structural work (AWS D1.2) in this introductory code explainer. Use these as educational touchpoints; always verify against the relevant standard, WPS/PQR, and your alloy/temper.
Shop procedures and temperature cues you can trust
- Melting and pouring in casting: Most foundry practice uses superheat—raising melt temperature above the alloy’s liquidus—to improve fluidity and feeding. The exact setpoint depends on alloy, hydrogen control, mold material, and shop SOPs. For A356, confirm pouring recommendations in an alloy datasheet or trusted handbook rather than copying internet numbers.
- Brazing vs. welding: Because alloys soften before they fully melt, fixtures and heat sinks help support parts when brazing near the solidus. Consistent torch technique or controlled induction profiles limit time in the mushy zone.
- Elevated-temperature service: Many aluminum alloys start losing significant strength above roughly 150–200 °C (300–392 °F). The Aluminum Association notes this threshold clearly in its publication on fire safety and elevated temperatures; see the official guidance in Fire Safety of Aluminum & Its Alloys (2020). Treat these temperatures as caution zones rather than hard limits and consult alloy-specific data for design decisions.
What about comparisons to other metals for intuition? Copper melts near 1084 °C (1983 °F) and typical steels around 1425–1540 °C (2597–2800 °F), vastly higher than the melting point of aluminum. For concise comparison charts, see industry references such as the Metal Supermarkets melting-points overview alongside the NIST value for aluminum cited earlier.
Troubleshooting for small shops
- If the edge collapses or sags near the toe of the weld: You’ve likely drifted into the mushy zone at the edge (near solidus). Increase travel speed, reduce dwell, switch to stringers, add chill bars or heat sinks, and verify fit-up to shorten arc time.
- If weld strength seems low after a clean-looking bead: Suspect HAZ softening in heat-treatable alloys. Review heat input, interpass practices, and whether the procedure preserves temper. Consider test coupons to validate.
- If porosity and lack of fusion appear together: Improve cleaning, wire/rod storage, and gas shielding. Both flaws increase rework time and heat exposure, compounding HAZ damage risk.
Mini-cases to make it concrete:
- TIG on 1/8 inch 6061 plate: Keep arc tight and move. Favor a smaller cup with good gas coverage and a brisk travel speed. If the HAZ turns wide and matte, you’re likely overaging; back off on amperage or use pulsed current to limit average heat.
- Rim repair by brazing: The part can look fine but be mechanically weak if you linger near the solidus. Use heatsinks and controlled, minimal flame; stop at the first adequate flow and let it cool under restraint.
- Small A356 casting batch: Superheat improves fill, but the optimal temperature depends on your melt practice and mold. Rather than chase a single “best” temperature from the web, record trials near your qualified procedure and watch for surface finish, shrink, and hydrogen defects, adjusting within the datasheet’s guidance.
Safety reminders for any heat work on aluminum: Wear appropriate PPE, ensure ventilation, keep a dry work area free of moisture near molten metal, and follow local codes and standards for welding and foundry operations.
Appendix and further reading
Unit conversions you’ll use often:
°F = (°C × 9/5) + 32
°C = (°F − 32) × 5/9
K = °C + 273.15
Authoritative references cited in this guide:
- Pure-element constant for aluminum’s melting point: see the NIST Chemistry WebBook aluminum entry (Tfus ≈ 933.45 K → 660.32 °C / 1220.58 °F).
- Solidus–liquidus behavior and eutectics in Al–Si: overview in ASM’s Alloy Phase Diagrams content on eutectic systems.
- Alloy ranges and properties: ASM/MatWeb 5052-O, ASM/MatWeb 6061-T6, and ASM Handbook A356 chapter.
- Elevated-temperature strength loss context: Aluminum Association — Fire Safety of Aluminum & Its Alloys (2020).
- HAZ behavior and shop controls: ESAB explainer on aluminum HAZ and related training posts referenced above.
Final note: Use the melting point of aluminum as a starting point, then work from your alloy’s solidus–liquidus range and your procedure qualifications. That’s how you keep parts straight, strong, and in one piece.





