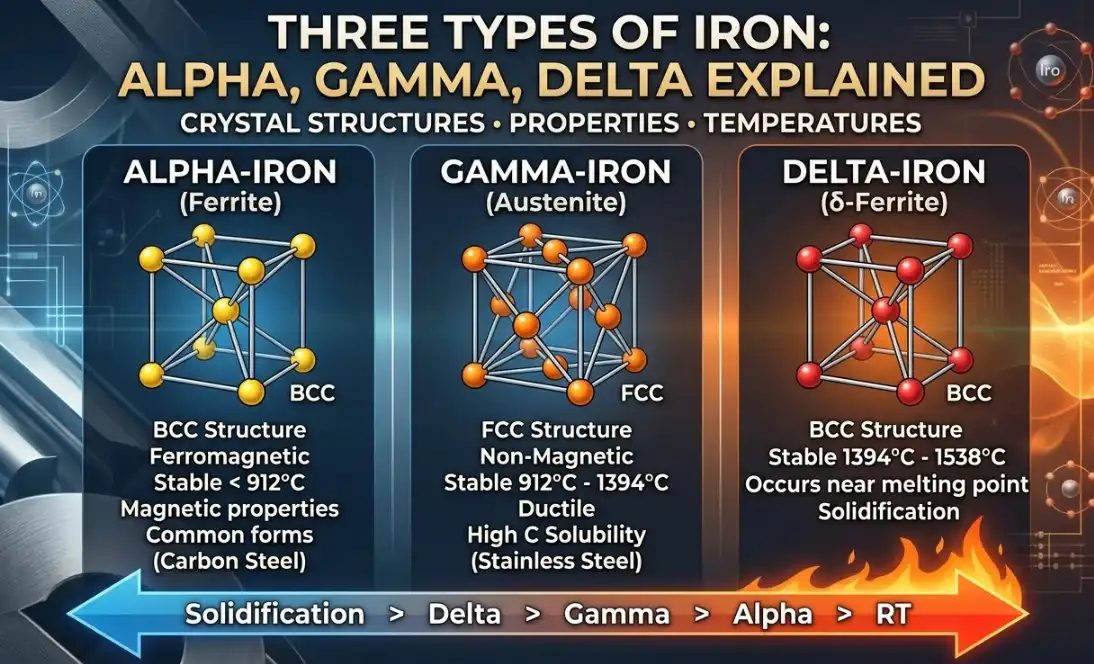
When engineers say the three types of iron, they mean the three allotropic forms of pure iron at ambient pressure: alpha-iron (α-Fe, ferrite), which has a body-centered cubic (BCC) crystal structure and is stable from room temperature up to about 912 °C; gamma-iron (γ-Fe, austenite), which is face-centered cubic (FCC) and stable from roughly 912 °C to about 1394 °C; and delta-iron (δ-Fe), a high-temperature BCC phase stable from approximately 1394 °C to the melting point near 1538 °C. A concise, authoritative summary of these phases—their names, crystal structures, and temperature intervals—appears in Encyclopaedia Britannica’s overview of iron, which describes iron’s allotropy in standard physical metallurgy terms: Britannica’s article on iron.
Quick reference: the three types of iron
Alpha iron — ferrite — BCC
- What it is: α-Fe is the BCC phase of pure iron. It is ferromagnetic below its Curie temperature (~770–771 °C) and becomes paramagnetic above that, while remaining BCC until ~912 °C. For the magnetic transition itself, see the explanation of the Curie point in Britannica’s Curie point entry.
- Why it matters: Ferrite is the soft, ductile matrix present at ambient temperature in many steels. Its limited carbon solubility drives the formation of ferrite and pearlite during slow cooling of austenite. The distinction between the magnetic Curie transition and the structural α→γ transition is outlined clearly in the University of Cambridge Phase Transformations site.
Gamma iron — austenite — FCC
- What it is: γ-Fe is the FCC phase of pure iron, stable from ~912 °C to ~1394 °C. Austenite dissolves far more carbon than ferrite because of its FCC geometry and larger, more numerous octahedral interstitial sites—one of the core reasons it’s central to steel heat treatment.
- Why it matters: Most heat treatments rely on austenitizing—heating into the γ field—so carbon (and alloying elements) enter solid solution. On cooling, the austenite decomposition route (to ferrite/pearlite, bainite, or martensite) sets the final properties.
Delta iron — high-temperature BCC
- What it is: δ-Fe is BCC again, reappearing from ~1394 °C up to iron’s melting point (~1538 °C). While brief in many process schedules, it matters in casting and at the very high-temperature end of steelmaking.
- Why it matters: Knowing δ-Fe helps interpret solidification paths and the upper reaches of the iron–carbon diagram.
Note: The Curie temperature (~770–771 °C) marks a magnetic transition in α-iron (ferromagnetic to paramagnetic). The crystal structure remains BCC up to ~912 °C, where the structural transition to FCC (γ-Fe) occurs. Cambridge’s Phase Transformations materials provide a succinct distinction between magnetic and structural transitions.
Why the three types of iron matter in steelmaking and heat treatment
If you work with steels, you spend your life managing austenite and its transformations. Heating a plain carbon steel above the upper critical temperature (Ac3 for hypoeutectoid grades) fully converts ferrite/pearlite to austenite. Holding in the austenite range allows carbon to homogenize. Cooling then determines what forms:
- Slow furnace cooling produces ferrite and pearlite under near-equilibrium conditions.
- Intermediate, isothermal holds can produce bainite.
- Fast quenching creates martensite.
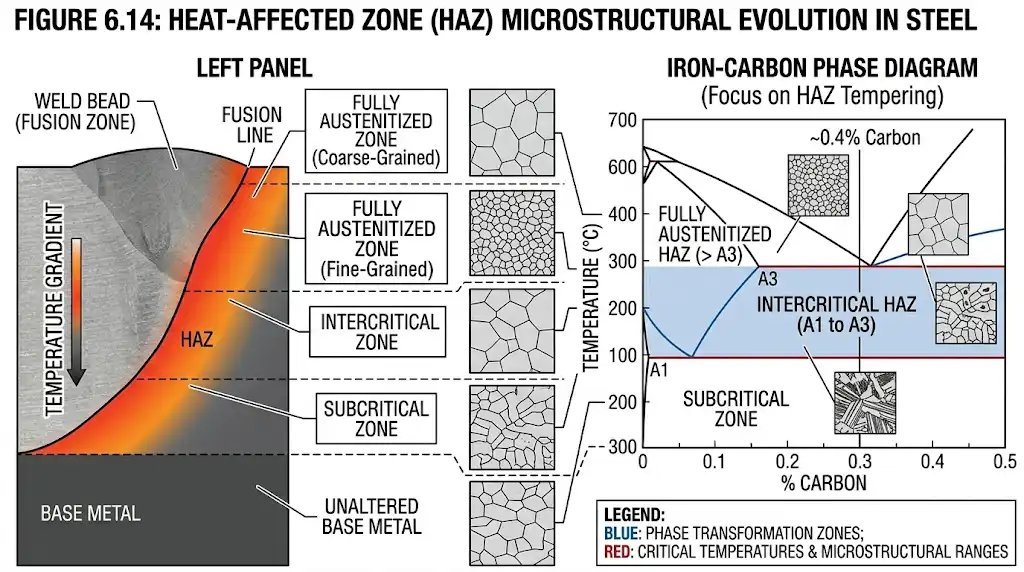
In welding, the heat-affected zone (HAZ) shows the same phase logic at smaller scales. Regions heated above Ac3 fully austenitize; the intercritical HAZ (between Ac1 and Ac3) partially austenitizes; the subcritical HAZ remains ferritic/pearlitic but may temper any pre-existing martensite. For a practitioner’s overview of HAZ sub-zones and expected transformations in carbon steels, see TWI’s primer on the heat-affected zone.
For orientation (values are grade-dependent and must follow standards), hypoeutectoid plain carbon steels are often austenitized roughly 30–50 °C above Ac3—commonly around ~780–900 °C depending on carbon and alloying—while many hypereutectoid grades are austenitized between Ac1 and Acm (roughly ~750–830 °C) to dissolve enough carbides without excessive grain growth. Authoritative, grade-specific guidance is compiled in ASM International resources and relevant standards; always verify for the particular specification.
Carbon in BCC vs FCC and the iron–carbon diagram
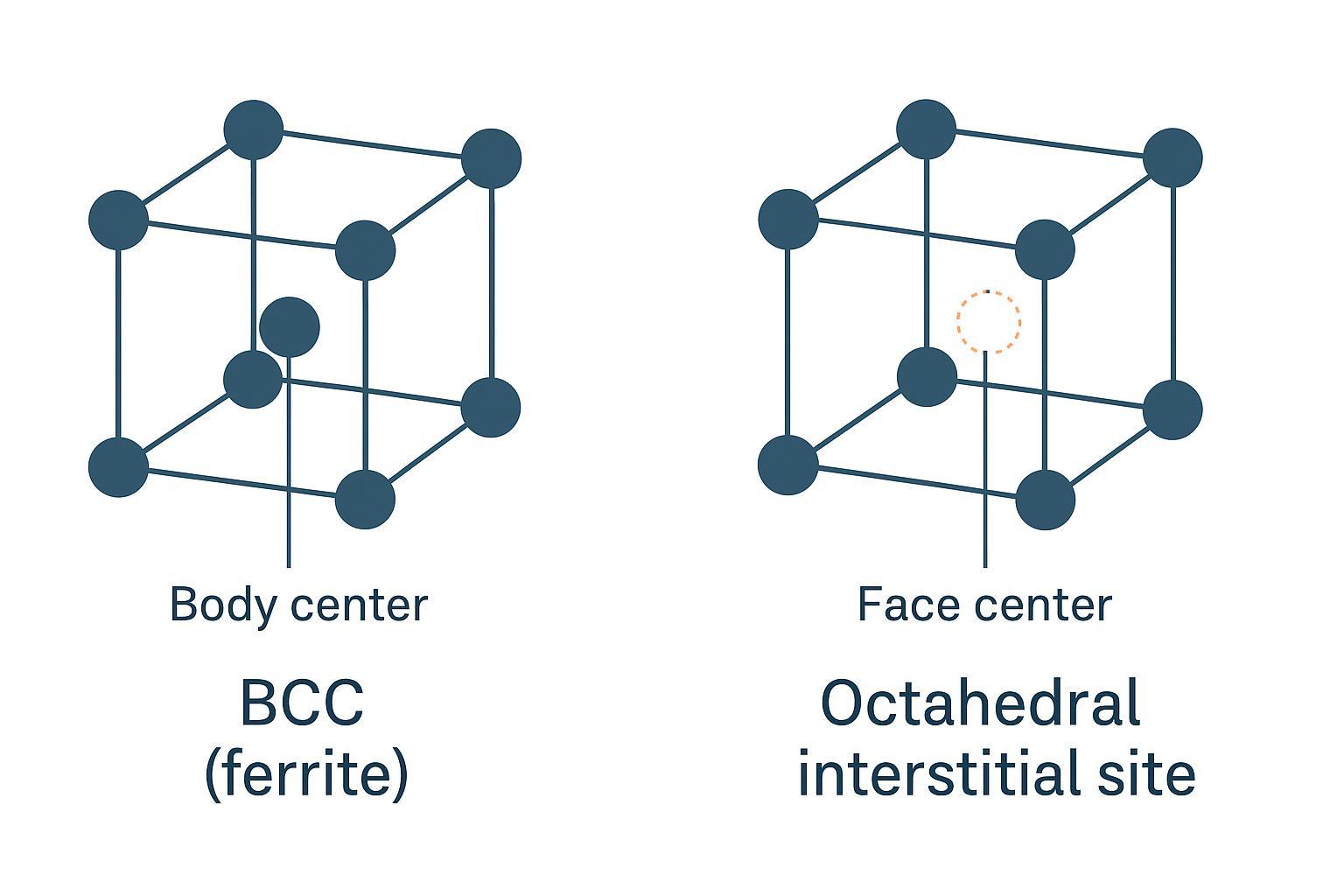
Why does austenite dissolve more carbon than ferrite? Think of the lattice like a parking garage for interstitial atoms: FCC has more and roomier “spots” (octahedral sites) than BCC, so squeezing carbon in causes less strain. As a result, γ-Fe can hold orders of magnitude more carbon than α-Fe before carbide precipitation becomes favorable. The University of Cambridge Phase Transformations site explains interstitial site sizes in BCC versus FCC and connects them to solubility in a clear, engineering-friendly way.
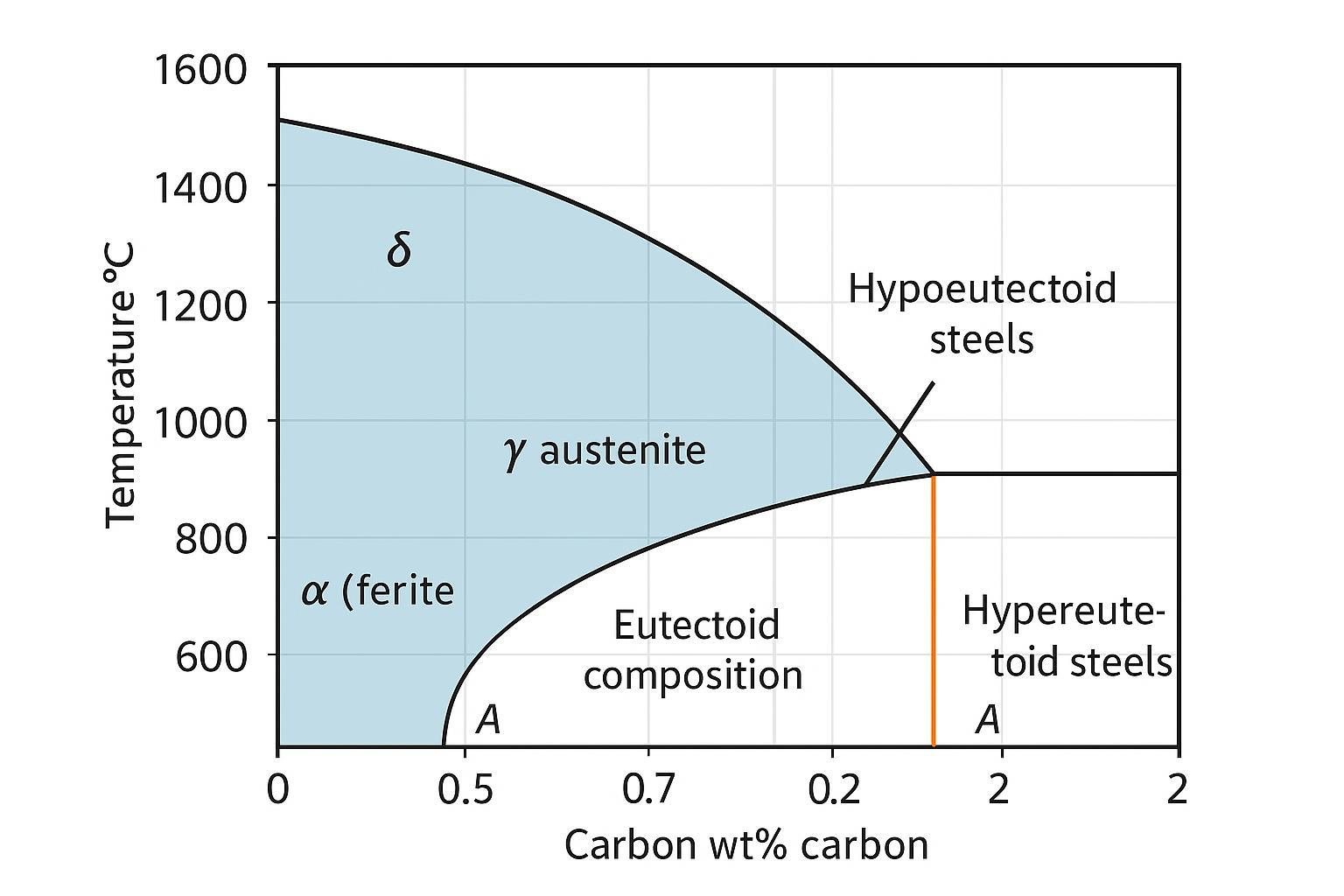
An anchor point on the iron–carbon diagram is the eutectoid reaction near ~0.77 wt% C at about 727 °C: on cooling, austenite transforms to a lamellar mixture of ferrite and cementite called pearlite. For a concise overview of the diagram and its invariant points, see the iron–carbon phase diagram article, which aligns with textbook values and provides a familiar schematic for quick reference. Orientation-only austenitizing guidance by steel family should be taken from standards or ASM texts because alloying shifts the lines.
| Phase (pure Fe) | Common name | Crystal structure | Approximate temperature range (1 atm) | Key notes |
|---|---|---|---|---|
| α-Fe | Ferrite | BCC | Room temperature to ~912 °C | Curie ~770–771 °C (magnetic transition only) |
| γ-Fe | Austenite | FCC | ~912–1394 °C | Much higher carbon solubility than ferrite |
| δ-Fe | Delta iron | BCC | ~1394 °C to ~1538 °C | Exists just below melting |
Clarification: phases versus product classes
Outside metallurgy circles, you’ll sometimes hear the three types of iron listed as wrought iron, cast iron, and steel. Those are alloy/product categories distinguished mainly by carbon content and processing, not the allotropic phases of pure iron.
- Wrought iron: Historically very low-carbon iron worked mechanically and containing slag inclusions; largely superseded by modern low-carbon steels.
- Steel: Fe–C alloys typically up to about 2 wt% carbon (often much less in practice), with microstructures of ferrite, pearlite, bainite, or martensite depending on processing.
- Cast iron: Fe–C alloys typically above ~2.0–2.1 wt% carbon, forming graphite (gray, ductile) or cementite (white iron) depending on composition and cooling.
Authoritative definitions and context are provided in Britannica’s article on iron, which links to related entries on steel and cast iron.
Frequently seen notations and terms
Common shorthand and symbols in standards and textbooks include: α-Fe (ferrite) for BCC iron at room temperature; γ-Fe (austenite) for the high-temperature FCC phase; δ-Fe for the high-temperature BCC phase; Ac1 and Ac3 for critical transformation temperatures on heating (Ar1 and Ar3 on cooling); Fe3C for cementite; and the names of transformation products—pearlite, bainite, martensite—that form from austenite during cooling (these are not allotropes of pure iron). See how compact that vocabulary is when you tie it back to the three types of iron?
Putting the three types of iron to work
Here’s the deal: once you connect the three types of iron to the iron–carbon diagram, you can read heat-treatment specifications with confidence and predict weld HAZ outcomes more reliably. Need to refine grain size? Control your austenitizing temperature and time. Chasing a balance of strength and toughness? Tune the austenite decomposition path with cooling rate and isothermal holds. And if you ever hear someone say “steel is one of the three types of iron,” you’ll know exactly how to clear up the confusion in a single sentence.