You may think stainless steel does not rust. It still has iron inside. Iron reacts with water and oxygen. This makes stainless steel rust. Chromium in stainless steel makes a thin shield. You cannot see this shield. The shield stops rust most of the time. Harsh chemicals or scratches can break the shield. Higher grades like 316 resist rust better than 304 or 18/8. They usually cost about 40% more.
| Stainless Steel Grade | Corrosion Resistance | Typical Cost Difference |
|---|---|---|
| 304 | Moderate | Baseline |
| 316 | High | ~40% more expensive |
| 18/8 | Moderate | Similar to 304 |
Key Takeaways
- Stainless steel can rust because it has iron in it. You should keep it clean and make sure there are no scratches.
- Pick the right stainless steel grade for where you use it. Grade 316 works best in places with salt or lots of chemicals.
- Clean it often with gentle soap and water. This helps get rid of dirt and things that cause rust.
- Use special treatments like passivation or coatings. These help stop rust and make stainless steel last longer.
- Do not use steel wool or rough pads. They can scratch the surface and hurt the chromium layer that protects it.
Corrosion Resistance of Stainless Steel
Chromium and the Protective Layer
You might ask why stainless steel does not rust as fast as regular steel. The main reason is chromium. If stainless steel has enough chromium, it gets a strong shield. This shield protects it from rust. Chromium mixes with oxygen in the air. This makes a thin oxide layer on top of the steel. You cannot see this layer. It keeps water and air away from the iron inside.
- When chromium is more than 10.5%, the oxide layer forms and protects the steel.
- You need at least 12% chromium to keep the oxide layer strong. If there is less chromium, rust can start.
- Most austenitic stainless steel alloys have 10% to 20% chromium. Ferritic stainless steel alloys can have up to 29% chromium.
Austenitic stainless steels typically contain about 16–26% chromium; for example, grade 304 commonly has ≈18–20% Cr and 8–10.5% Ni, while grade 316 typically has ≈16–18% Cr, 10–14% Ni and 2–3% Mo, according to industry datasheets and standards summaries such as WorldStainless grade sheets and Outokumpu’s austenitic overview.
The oxide layer is very important. If you scratch or hurt the surface, chromium helps the layer fix itself. This self-healing makes stainless steel last longer in hard places.
Role of Nickel and Molybdenum
Nickel and molybdenum also help stainless steel fight rust. Nickel makes the steel stronger and easier to bend. It helps the oxide layer stay in place. Molybdenum gives extra help, especially where there is salt or chemicals.
Molybdenum makes stainless steel better at stopping pitting rust. It also helps against crevice rust and wear. More molybdenum means even better pitting rust resistance than more chromium. Molybdenum also helps the oxide layer stay strong.
Nickel and molybdenum work together to make stainless steel even better at stopping rust. They make the oxide layer stronger and keep your stainless steel safe.
Many scientists have studied how these metals protect stainless steel. Some important studies are by Sadough Vanini, Baba, Stewart, Wijesinghe, Tian, Ilevbare, Leckie, Sugimoto, and others. These studies show how the oxide layer and other metals stop rust in different places.
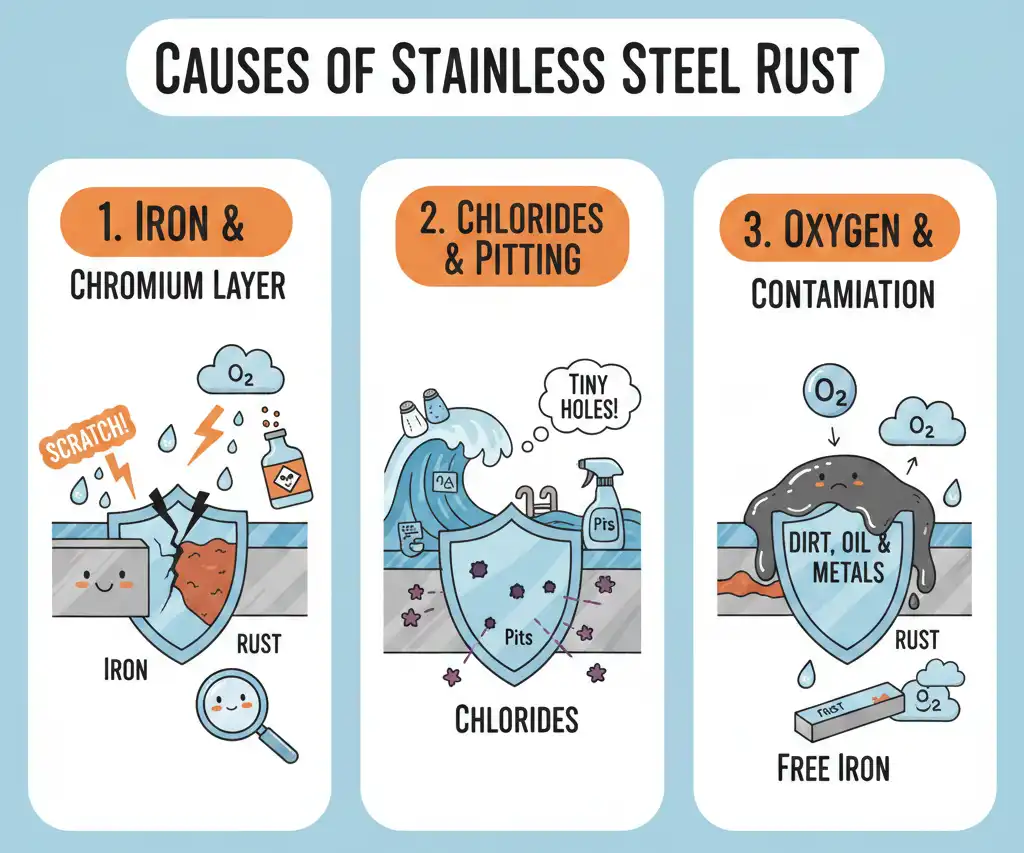
Causes of Stainless Steel Rust
Iron Content and Oxidation
Stainless steel can rust because it has iron inside. Iron and chromium are the main parts of stainless steel. Chromium makes a thin layer on top. This layer is invisible. It keeps water and air away from the iron. If you scratch or break this layer, iron gets exposed. Iron will rust when it touches water and air.
- The chromium layer can break from scratches, strong chemicals, or heat.
- If the layer breaks, water and air reach the iron and rust starts.
- Chromium mixes with oxygen to make a shield, but if the shield gets damaged, rust can happen fast.
Free iron on the outside is different from iron inside stainless steel. Free iron rusts faster when it meets water and air.
Chlorides and Pitting Corrosion
Chlorides are found in salt, seawater, pool water, and tap water. These chemicals can hurt the protective layer on stainless steel. When chlorides break the shield, they make tiny holes called pits. This is called pitting corrosion. You might see small brown spots or rough areas.
- Chloride ions can break the oxide layer and start pitting.
- The chance of pitting depends on the stainless steel grade, how much chloride there is, the temperature, and the surface finish.
- Seawater and pool water can make rust happen faster because chlorine is very strong.
- Strong cleaning chemicals can also break the shield and cause rust.
If you use stainless steel in kitchens, bathrooms, or near the sea, watch out for these problems. Even a little salt or cleaner can hurt the steel over time.
Oxygen and Surface Contamination
Oxygen helps the chromium layer stay strong. The layer needs oxygen to fix itself. If dirt, oil, or other metals cover the surface, the layer cannot heal. This makes rust easier to start.
- Free iron on the surface can mix with water and air and make rust spots.
- Rust looks bad and can make people think the metal is not stainless steel.
- Rust can get into food, water, or other things that touch the steel.
If you see rust on stainless steel, it often means the surface got dirty with free iron or other stuff. Cleaning and keeping the surface clean helps stop rust.
The environment matters too. High heat can make the protective layer weak. Changes like humidity or pollution can make rust happen faster. Scratches, bumps, or fingerprints can hurt the surface and help rust start.
- Strong chlorides, very hot or cold temperatures, and harsh chemicals often cause rust at home or in factories.
- Scratches or damage let air and water reach the steel and start rust.
If you know what causes rust, you can keep your stainless steel safe.
Preventing Rust on Stainless Steel
Cleaning and Maintenance
You can keep stainless steel shiny by cleaning it often. Cleaning removes dirt, salt, and other things that cause rust. In places like kitchens or near the sea, clean and rinse the surface a lot. This gets rid of chlorides and other bad stuff. Do not let water sit on the surface. Water left on stainless steel can make rust start. Check and clean small spaces because dirt and salt can hide there.
- Clean with warm water or a mild soap.
- Use alcohol or acetone to get rid of oil and grease.
- For rust stains, use a cleaner made for stainless steel with nitric acid.
- Wipe off fingerprints with glass cleaner or a special cleaner.
Tip: Always rinse with water that has little chloride after cleaning. This helps stop chemicals from hurting the surface.
Safety & compliance note — do not attempt industrial acid work at home: Always prioritize safer DIY options (for example, citric-acid passivation) or hire a professional. Wear chemical-resistant gloves, safety goggles, and long sleeves; work outdoors or with strong ventilation. Never use steel wool or other iron-containing abrasives (they embed free iron). Do not use concentrated nitric acid at home, and never pour strong acids down drains—follow local hazardous-waste rules or contact a waste facility. See citric-acid passivation guidance and passivation safety for details.
| Contaminant Type | Recommended Cleaning Method |
|---|---|
| Dust or mild dirt | Wipe with warm water or mild detergent |
| Oil and grease | Use organic solvents (e.g., alcohol or acetone) |
| Rust stains | Apply a nitric acid-based stainless steel cleaner |
| Embedded iron particles | Perform acid pickling or passivation |
| Fingerprints | Use a glass cleaner or a specialized surface cleaner |
Choosing Rust Resistant Grades
Picking the right stainless steel helps stop rust. Some types work better in tough places. If you live by the ocean or use stainless steel in a pool, you need a type that can handle salt and water.
| Stainless Steel Grade | Chromium Content | Molybdenum Content | Key Features |
|---|---|---|---|
| Grade 316 | 16% | Up to 3% | High resistance to saltwater, iodides, and acids; longer lifespan in marine environments. |
| Grade 304 | 18% | 0% | Good resistance to caustic substances; prone to pitting in saltwater. |
- 304 stainless steel works for most things. It fights rust well and is used in cars and storage.
- 316 stainless steel is best for places with lots of salt or chemicals. It is used in food factories and near the sea.
- 316L stainless steel has less carbon. It is easier to weld and works well where there is a lot of rust.
Note: Tests like salt spray and passivation show these types fight rust in different ways. Picking the right type helps you stop rust and makes your things last longer.
PREN example (quick, practical): PREN (Pitting Resistance Equivalent Number) is commonly calculated as PREN = %Cr + 3.3×%Mo + 16×%N. Using typical mid‑range compositions gives a quick comparison:
- 304 (typical): Cr ≈ 18.5%, Mo = 0%, N ≈ 0.08% → PREN ≈ 18.5 + 0 + 1.28 = ~19.8
- 316 / 316L (typical): Cr ≈ 17.0%, Mo ≈ 2.5%, N ≈ 0.08% → PREN ≈ 17 + 8.25 + 1.28 = ~26.5
Interpretation: a higher PREN indicates better pitting resistance—316’s PREN is roughly 30–35% higher than 304’s, which helps explain its superior performance in chloride environments. PREN is a screening metric; for critical uses consult manufacturer datasheets or CPT (crevice/pitting tests) under expected service conditions.
Avoiding Surface Damage
You can stop rust by keeping the surface safe. The thin chromium oxide layer on top keeps rust away. Scratches or cuts can break this layer. When the layer breaks, air and water touch the steel. This makes rust start faster. Even small scratches can let rust begin.
Hard bumps or rubbing can also hurt the surface. If the damage is deep, it lets water and air reach inside. This makes rust more likely and can make the steel weak.
To keep your stainless steel safe:
- Be careful not to scratch it.
- Do not use steel wool or rough pads to clean.
- Pickling can clean stains and fix the surface. This uses acid to clean off dirt and heat marks.
- After pickling, rinse with water that has little chloride and use passivation. Passivation helps make a strong shield.
Remember: Keeping the surface clean and smooth helps the shield stay strong. This is the best way to keep your stainless steel from rusting.
Rust Resistant Treatments
You can help stainless steel last longer with special treatments. These treatments stop rust and make cleaning easier. There are three main ways: electropolishing, passivation, and protective coatings. Each one gives extra help and keeps stainless steel looking nice.
Electropolishing
Electropolishing uses electricity and a liquid bath to clean the surface. The stainless steel goes into a special solution. Electricity is used to remove tiny bumps and dirt. This makes a smooth and shiny surface. It also helps build a strong oxide layer. The oxide layer protects against rust.
- Electropolished surfaces can fight rust much better than untreated ones.
- The shiny finish is easy to wipe clean.
- Electropolishing changes the surface itself, not just adds something on top.
Tip: Electropolishing works well for things that must stay very clean, like medical tools or food machines.
Passivation
Passivation is a chemical way to make stainless steel resist rust better. You use acid, like nitric or citric acid, to clean the surface. This step takes away things that can cause rust. After passivation, the surface is less likely to rust.
- Nitric acid passivation removes tough dirt.
- Citric acid passivation is safer for the earth.
- Passivation does not hurt the metal or make it weaker.
Studies show passivation helps stainless steel last longer in tough places, like factories or airplanes. If rust does show up, passivation makes it easier to clean off.
Safety & compliance
Pickling and chemical passivation involve strong acids and should only be done in a controlled, professional setting. Refer to ASTM A380 and ASTM A967 for industry procedures and verification. Minimum PPE includes acid-resistant gloves, splash goggles or face shield, acid-resistant apron, and appropriate respiratory protection; avoid enclosed or poorly ventilated areas. Do not neutralize or pour acid waste down drains—follow local hazardous-waste rules or return waste to a licensed processor.
| Passivation Method | Main Benefit | Typical Use Case |
|---|---|---|
| Nitric Acid | Strong rust removal | Industrial equipment |
| Citric Acid | Eco-friendly, safe | Food and medical tools |
| Laser Passivation | Reduces pitting corrosion | High-tech applications |
Protective Coatings
Protective coatings add another layer to stop rust. You can use paint, spray, or special films. These work best on good stainless steel, like 316L. Coated 316L stainless steel fights rust better than coated 304.
- Coatings slow down rust and make cleaning easier.
- You can pick coatings for different jobs, like outside or in chemical plants.
Note: Using coatings and picking the right stainless steel type gives you the best protection and longest life.
These treatments save money and time by lowering repairs and replacements. They help the earth too, because stainless steel lasts longer and you do not need to make new parts as often.
You have learned that stainless steel rusts when the chromium layer gets hurt or faces tough things. The table below lists the main causes:
| Factor | Effect on Rust Formation |
|---|---|
| Damage to Chromium Layer | Makes iron more likely to rust |
| Chloride Ions | Breaks down the protective layer |
| Water and Oxygen | Start the rust process |
To keep your things safe, try these ideas: Clean and dry the surface a lot. Use coatings or passivation for more safety. Choose the right grade for where you use it.
Taking care of your stainless steel helps it last longer and look bright.
References & Further Reading
- For industry passivation and cleaning practice, see ASTM A380/A380M (Cleaning, Descaling, and Passivation) and ASTM A967/A967M (Chemical Passivation Treatments). These are the canonical procedures used by fabricators and finishers.
- For laboratory corrosion testing (pitting/crevice), consult ASTM G48 (Pitting and Crevice Corrosion Tests).
- For practical corrosion guidance from industry bodies, see AMPP/NACE technical resources on corrosion control.
- Market context for grade pricing: 316 is typically priced higher than 304—commonly in the ~15–40% premium range (most often ~20–30%), but the actual premium depends on molybdenum and nickel commodity costs, product form and finish (sheet, bar, tube), regional supply, and order size; see stainless price trackers and market reports such as MEPS world stainless prices or distributor market notes.
(Note: standards pages are paywalled; cite the ASTM/AMPP landing pages for canonical references.)
FAQ
Why does my stainless steel rust even indoors?
You may see rust if the surface gets dirty or scratched. Water, cleaning chemicals, or salt can break the protective layer. Always keep your items clean and dry.
Can I remove rust from stainless steel at home?
Yes, you can use a cleaner made for stainless steel. For small spots, try baking soda and water. Rinse and dry the surface after cleaning.
Does stainless steel rust in saltwater?
Saltwater can cause rust faster. Salt breaks the protective layer and starts pitting. Use higher grades like 316 for the best results near the sea.
How can I stop fingerprints and stains?
Wipe surfaces with a soft cloth. Use a glass cleaner or a special spray for stainless steel. Clean often to keep your items shiny and free from marks.