The melting point of iron at normal atmospheric pressure is 1,538 °C (2,800 °F), which is 1,811 K. That single number applies to pure iron (the element Fe). In the real world, though, most of what we heat and shape is steel—iron plus carbon and other elements—which melts over a range instead of at one sharp temperature. Knowing the difference helps you choose safer, smarter windows for forging, casting, and welding.
According to the reference entry on iron in Encyclopaedia Britannica, pure iron “melts at 1,538 °C (2,800 °F),” a value that aligns with standard handbooks; the same value is listed in the NIH’s PubChem periodic table as 1,811 K. See the authoritative summaries in the Encyclopaedia Britannica article on iron and the PubChem periodic table melting point page:
- Pure iron value and properties in the Encyclopaedia Britannica summary: see the iron element overview in the article titled Iron | Element.
- Kelvin alignment and tabular data in the PubChem periodic table’s melting point entry for Fe.
To use this number well, let’s translate it into practical guidance you can trust.
What the melting point of iron really means (and why steel is different)
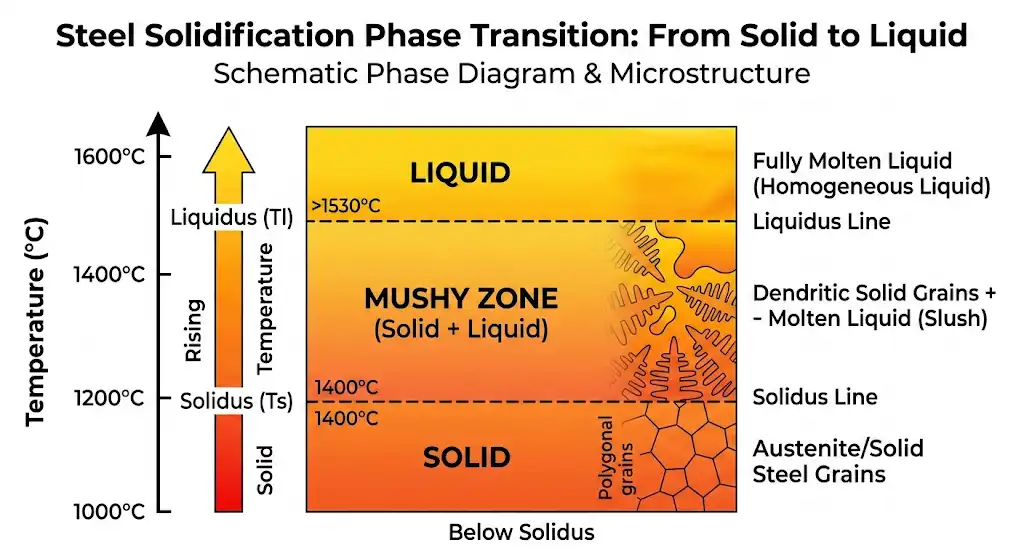
For a pure substance like elemental iron, “melting point” is the temperature where solid and liquid coexist in equilibrium at a given pressure (here, 1 atm). That’s our 1,538 °C benchmark for pure Fe. Steel, however, is not pure iron; it’s an alloy. Alloys don’t melt at a single temperature. Instead, they transition from fully solid to fully liquid over a span called the melting range. Two handy terms define that span:
- Solidus: below this temperature, the alloy is fully solid.
- Liquidus: above this temperature, the alloy is fully liquid.
Between those two is a semi-solid “mushy zone,” where both solid and liquid phases exist. Carbon and other alloying elements generally lower the temperatures at which steel begins to melt and widen that mushy zone. Introductory metallurgy references and ASM Handbooks discuss how the iron–carbon system sets these boundaries and why composition matters to melting behavior (see ASM’s solidification and forging chapters for foundational explanations).
Think of it like a snow cone on a warm day: for a while it’s part ice, part syrupy liquid. That in-between state is your mushy zone. You wouldn’t try to build a snowman out of a snow cone; by the same logic, you shouldn’t try to forge or weld in the mushy zone.
Quick glossary for beginners
Solidus — The highest temperature at which an alloy is still fully solid.
Liquidus — The lowest temperature at which an alloy is fully liquid.
Melting range — The span between solidus and liquidus; alloys pass through this interval rather than flipping from solid to liquid at one point.
Eutectic — A special composition where melting and solidifying happen at a single temperature lower than neighboring compositions; useful idea in iron–carbon alloys but beyond what most beginners need day to day.
The melting point of iron in practice: forging, welding, and casting
Here’s the deal: nearly all practical metalwork is done well below the liquidus to avoid loss of shape, cracking, and safety hazards. Your targets depend on the specific material, code requirements, and the job at hand. Always check the data sheet and applicable standard for the exact alloy.
Forging: stay in the safe, workable hot-solid window
For most carbon and low-alloy steels, common finish-forging temperatures fall around 980–1,095 °C (1,800–2,000 °F). Authoritative forging references consolidate these ranges and caution against overheating, which can cause defects such as hot shortness. See the forging ranges and cautions summarized in the ASM Handbooks chapter on forging of specific metals and alloys.
Practical pointers for students and hobbyists:
- Reheat early rather than pushing steel too cool; working too low raises cracking risk, while too hot risks surface burning or grain damage.
- Don’t rely only on color. Lighting, scale, and surface finish mislead. Use temperature-indicating crayons, a contact thermocouple, or a calibrated IR pyrometer with emissivity set for hot steel.
- Keep tools and anvils appropriately preheated for heavy work so they don’t chill your stock on contact.
Welding: preheat and interpass control—not “how hot to melt steel”
In welding, you rarely chase the melting point of iron; instead, you control how fast the welded region heats and cools. Preheat and interpass temperatures are selected to let hydrogen diffuse out and to avoid a brittle heat-affected zone (HAZ). A clear, beginner-friendly overview of why preheat matters is available from The Welding Institute; for how ranges are calculated in structural work, consult the American Welding Society’s explanations related to AWS D1.1 methods. See the accessible primer on why preheat is used from The Welding Institute and the overview article discussing preheat and interpass methods associated with AWS D1.1.
Student takeaways:
- Typical preheat for many low-carbon structural steels can range from ambient to roughly 50–150 °C (120–300 °F), but the correct value depends on carbon equivalent, thickness, restraint, and consumable hydrogen level. Always defer to the applicable code/table or engineering procedure.
- Monitor interpass so the joint doesn’t get too hot between passes; over-tempering can wreck toughness.
- Record actual temperatures; don’t guess. Use temperature crayons, contact thermocouples, or calibrated IR with a dull, consistent surface to improve readings.
Casting: melting vs. pouring (superheat)
Foundries typically pour metal a bit hotter than its melting range to stay fully liquid and fill molds. That extra headroom above the liquidus is the superheat. For steels, beginner-focused design education notes that superheats on the order of ~100–200 °F (≈56–111 °C) are common starting points, then tuned by alloy, section size, and mold complexity. See the casting design education guidance from the Steel Founders’ Society of America, which explains how alloy characteristics affect pouring choices and why “more heat” isn’t always better.
Principles that keep you safe and successful:
- Enough superheat to flow and fill; not so much that oxidation, gas pickup, or refractory attack skyrockets.
- Uniform temperature within the melt matters as much as the absolute number; tight control reduces defects.
- In school labs, only melt and pour under qualified supervision and written procedures.
How scientists and shops measure melting and temperature
In research labs, melting and solidification are determined by thermal analysis methods such as differential scanning calorimetry (DSC) or differential thermal analysis (DTA). Calibrating with pure-metal standards is essential, and the “extrapolated onset” of a melting peak is often used to identify critical temperatures. For plain-language best practices and calibration context, see NIST’s Special Publication 960-15 on DTA/DSC measurements.
In classrooms and shops, you’ll use practical tools:
- Temperature-indicating crayons (melt-on-contact sticks) to verify that a part reached a specific threshold.
- Contact thermocouples for direct readings on hot parts or in furnaces.
- Optical/IR pyrometers for non-contact measurements—be sure to set emissivity correctly and avoid shiny surfaces that read artificially low.
When welding, OSHA’s guidance on hazards and controls is a great overview of PPE, ventilation, and monitoring expectations for hot-work environments; it complements your temperature tools by reminding you how to protect yourself while you work.
Safety checklist (use in supervised labs and approved shops)
- Wear appropriate PPE: safety glasses under a face shield, welding helmet/hood as required, insulated gloves, flame-resistant clothing, and protective boots.
- Use local exhaust or fume extraction when welding, cutting, or pouring; avoid confined spaces without proper ventilation.
- Verify temperatures with instruments (crayons, thermocouples, calibrated IR). Don’t rely on color alone.
- Keep water away from molten metal; moisture can flash to steam and erupt violently.
- Follow your institution’s SOPs, data sheets, and applicable codes. If unsure, stop and ask a qualified supervisor.
Troubleshooting: common temperature-related issues
| Problem | Likely cause | What to try next |
|---|---|---|
| Steel cracks during forging | Working too cold; high strain at low temperature; pre-existing defects | Reheat sooner; reduce reduction per heat; confirm you’re within the recommended forging window for that grade |
| Burned surface or crumbling edges when forging | Overheating near the liquidus; oxidation | Lower the furnace setpoint; shorten soak time; work farther below liquidus; improve atmosphere control |
| Porosity in a casting | Too little superheat; turbulence; gas pickup | Increase superheat modestly; refine gating to reduce splash; improve melt cleanliness and de-gassing procedures |
| Incomplete mold fill | Viscosity too high (too cool); poor gating | Add controlled superheat; preheat molds as specified; adjust gating to maintain steady flow |
| Weld HAZ cracking | Inadequate preheat or high hydrogen | Calculate preheat per code; dry/low-hydrogen consumables; control interpass and heat input |
| Soft weld or poor toughness | Excessive interpass/over-temper | Reduce interpass; allow cooling within the specified range; verify procedure parameters |
Frequently asked questions
Does steel melt at the same temperature as pure iron?
No. Steels are alloys and melt over a range—usually below 1,538 °C—bounded by their solidus and liquidus. Composition (carbon, manganese, chromium, etc.) sets that range, which is why data sheets matter.
How hot should I make steel for forging?
It depends on the grade and section size, but finish-forging windows around 980–1,095 °C (1,800–2,000 °F) are common for many carbon and low-alloy steels according to consolidated forging references. Start with your alloy’s published guidance and stay well below liquidus.
Why not rely on color to judge temperature?
Color varies with lighting, scale, and surface condition. Instruments—temperature crayons, thermocouples, calibrated IR—are more reliable. Use color only as a rough cue, verified by a tool.
What’s the difference between melting and pouring temperature in casting?
Pouring temperature is typically set above the liquidus by a controlled “superheat” to keep the metal fully liquid and fill the mold. Too much superheat can increase oxidation and gas pickup.
Does pressure or atmosphere change the melting point of iron for my shop work?
At everyday shop pressures, the melting point of pure iron effectively stays the same; atmosphere mainly affects oxidation and cleanliness rather than the nominal melting temperature you’d use in beginner planning.
Sources and further reading
- The definitive melting point of pure iron is summarized in the Encyclopaedia Britannica article titled Iron | Element, which states 1,538 °C (2,800 °F). https://www.britannica.com/science/iron-chemical-element
- The NIH-backed PubChem periodic table lists the melting point of Fe as 1,811 K and aligns with the Celsius/Fahrenheit values used above. https://pubchem.ncbi.nlm.nih.gov/periodic-table/melting-point
- Consolidated forging ranges and cautions appear in the ASM Handbooks chapter Forging of Specific Metals and Alloys. https://dl.asminternational.org/handbooks/edited-volume/49/chapter/616018/Forging-of-Specific-Metals-and-Alloys
- Why welders use preheat and how interpass control protects toughness is explained in The Welding Institute’s introductory guidance. https://www.twi-global.com/media-and-events/insights/why-preheat
- For how ranges are determined in structural codes, see the American Welding Society’s overview on preheat and interpass related to AWS D1.1. https://www.aws.org/magazines-and-media/welding-digest/wd-june-2025-preheat-and-interpass/
- Casting design education materials from the Steel Founders’ Society of America discuss alloy characteristics and why controlled superheat matters. https://www.sfsa.org/subject-areas/education/information-for-casting-designers/alloy-characteristics-affect-casting-design/
- NIST Special Publication 960-15 outlines best practices for DTA/DSC when determining melting/freezing behavior and calibrations. https://nvlpubs.nist.gov/nistpubs/Legacy/SP/nistspecialpublication960-15.pdf
- OSHA’s welding hazards and solutions page summarizes key PPE and ventilation expectations for hot work. https://www.osha.gov/welding-cutting-brazing/hazards-solutions
Wrap-up: The melting point of iron—1,538 °C for pure Fe—is your anchor. Most steels start to soften and melt below that, over a range that depends on composition. If you respect that fact, verify temperatures with instruments, and work within conservative windows, you’ll forge, weld, and cast more safely and with fewer surprises. Stay curious, and always let the material data and the code or standard lead the way.