You might ask if there are non-conductive metals. In science, all metals let electricity pass through them. But some people call metals with very low conductivity non-conductive metals. It is important to know this because grounding keeps you safe from electric shock. When you use metals, grounding helps protect you. Grounding also stops fires from starting. If you know which metals have low conductivity, you can pick better metals for grounding in homes, cars, and factories.
- Grounding keeps you safe from electrical dangers.
- Grounding systems need metals that conduct electricity.
- Picking the right metal for grounding matters a lot.
Key Takeaways
- Non-conductive metals slow down electricity. This makes them safer for grounding in electrical systems.
- Grounding stops electric shocks and fires. You need to pick the right metal for safety. Bismuth, titanium, lead, and tungsten are non-conductive metals. Each has special properties.
- Testing methods like the eddy-current method help find non-conductive metals. This keeps things safe. You can look at color and weight to spot non-conductive metals. But testing is very important.
- Industries use non-conductive metals in safety devices, electronics, and aerospace. These metals help control heat and electricity.
- Always follow safety rules when working with metals. Picking the wrong metal can be dangerous. Knowing about metal conductivity helps you make good choices for electrical projects.
What Are Non-Conductive Metals
Definition
You might wonder what non-conductive metals are. In science, this means metals that do not let electricity move easily. These metals have a lot of resistance to electric current. No metal can stop all electricity, but some slow it down a lot. People call these metals non-conductive because they act more like non-conductive materials than normal metals. In factories and labs, workers use this word for metals that help with insulation or safety.
Scientific Meaning
Most metals let electricity move quickly. This is because metals have free electrons. These electrons jump from atom to atom and carry electric current. Some metals have a structure that makes it hard for electrons to move. If a metal has many impurities or a messy atomic pattern, it does not conduct electricity well. Scientists say these metals have high resistivity. You can use these metals in special jobs where you want to slow down electricity. That is why people sometimes call them non-conductive metals.
Industry Usage
Many industries use non-conductive metals for safety and protection. Workers use these metals to keep machines safe from electric shocks. They also use them to stop electrical noise in sensitive equipment. Sometimes, industries put special coatings on metals to make them even more non-conductive.
Here is a table that shows some common coatings and how people use them:
| Type of Coating | Application Description |
|---|---|
| Polyurethanes | Used for heat and electrical insulation, stopping overheating and electrical flow. |
| Epoxies | Makes a strong insulating layer on metal surfaces. |
| Ceramic coatings | Works as a thermal barrier, limiting heat and stopping failures. |
| Polymers | Used in many industrial jobs for insulation. |
You can find these coatings in cars, airplanes, and electronics. They help protect people and machines from harm. When you pick metals for grounding, you need to know if the metal is non-conductive or has a special coating. This helps you make safe choices and avoid accidents.
Metal Conductivity Explained
Atomic Structure
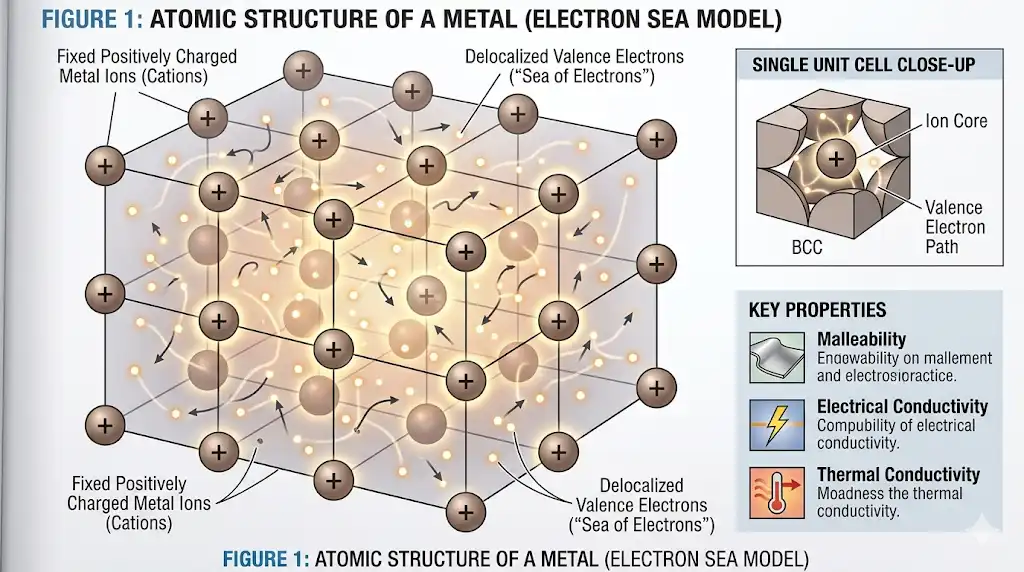
You might wonder why metals carry electricity and heat well. The answer is in how their atoms are arranged. Metals have atoms in a neat pattern called a crystal lattice. Some electrons, called valence electrons, can move around easily. These electrons do not stay with one atom. They move through the lattice and help with conductivity.
- Valence electrons are important for electrical conductivity in metals.
- Metals with loose outer electrons let them move more. This makes electrical conductivity and thermal conductivity higher.
- How atoms are packed in the lattice can change conductivity. If atoms are packed tighter, electrons cannot move as well. This lowers electrical conductivity and thermal conductivity.
So, atomic structure matters for both electrical conductivity and thermal conductivity. When electrons move freely, the metal conducts electricity and heat better.
Why Metals Conduct Electricity
You may ask why metals conduct electricity so well. The reason is how electrons act inside metals. Metals have many electrons near the Fermi level. These electrons can move when voltage is added. When you connect a battery, an electric field forms. This field pushes the free electrons. The electrons drift and make an electric current. This also helps with thermal conductivity because moving electrons carry heat.
- Electric current is electrons moving. Metals have many free electrons that move easily. This gives high electrical conductivity and thermal conductivity.
- The lattice lets electrons move without getting stuck.
- If temperature goes up, atoms vibrate more. This can slow electrons down. Electrical conductivity and thermal conductivity get lower.
- Impurities in the metal can block electrons. This also lowers electrical conductivity and thermal conductivity.
Think about silver and aluminum. Silver has fewer free electrons than aluminum. But silver’s electrons move easier. This makes silver better for electrical conductivity and thermal conductivity.
Limits Of Conductivity
Not all metals conduct electricity and heat the same. Some metals have much lower electrical conductivity and thermal conductivity. You can measure these limits with special units. The table below shows how some metals compare:
| Material | Minimum Conductivity (MS/m) | Thickness (mm) | Frequency (kHz) |
|---|---|---|---|
| TC4 Titanium | 0.5 | 0.8 | 1400 |
| 1060 Aluminum | 30 | 0.8 | 24 |
| TU1 Copper | 55 | 0.8 | 13 |
You can see TC4 titanium has much lower electrical conductivity and thermal conductivity than aluminum or copper. This means titanium slows down electricity and heat. When you pick metals for safety, you need to know these limits. Choosing a metal with low electrical conductivity and thermal conductivity can help in special jobs. It can block or slow down electricity and heat.
Note: Always check the electrical conductivity and thermal conductivity of a metal before using it in electrical systems. This helps you make safe and smart choices.
Examples Of Non-Conductive Metals
Bismuth
Properties
Bismuth stands out as one of the most non-conductive metals you can find. It has a silvery-white color with a pinkish tint. You will notice that bismuth feels heavy for its size. It does not rust easily and stays stable in air and water. Bismuth has a low melting point compared to other metals. It melts at about 271°C (520°F). This metal does not let electricity move through it quickly. Its atomic structure makes it act more like non-conductive materials than most metals. You can use bismuth in places where you want to slow down or block electric current.
Uses (e.g., fuses)
You will see bismuth used in safety devices. One common use is in electrical fuses. When too much current flows, the bismuth melts and breaks the circuit. This keeps you safe from electrical fires. Bismuth also appears in some types of solders. It helps lower the melting point, making the solder easier to use. You can find bismuth in medical tools and cosmetics because it does not react with skin. Its non-conductive nature makes it useful in special alloys and as a replacement for lead in some products.
Tungsten
Properties
Tungsten is a dense, gray metal. It has a very high melting point, which means it stays solid even at high temperatures. You will find that tungsten is hard and strong. It resists wear and does not bend easily. Tungsten does conduct electricity, but not as well as copper or aluminum. Here is how tungsten’s conductivity compares:
- Tungsten’s electrical conductivity: about 18.2 x 10^6 S/m
- Copper’s electrical conductivity: higher than tungsten
- Aluminum’s electrical conductivity: about 37.7 x 10^6 S/m, also higher than tungsten
This means tungsten slows down electric current more than copper or aluminum. You can use tungsten when you need a metal that handles heat but does not carry electricity as quickly.
Applications
You will see tungsten in light bulb filaments. It glows without melting because of its high melting point. Tungsten also appears in X-ray tubes and electrical contacts. Its low conductivity helps control the flow of electricity in these devices. You can find tungsten in aerospace and military equipment. It works well in places where you need strength and resistance to heat, but you do not want electricity to move too fast.
Lead
Properties
Lead is a soft, heavy metal with a dull gray color. It bends easily and feels dense in your hand. Lead does not conduct electricity as well as copper or aluminum. You can see how lead-based solders compare in this table:
| Solder Composition | IACS Score (%) |
|---|---|
| 15/85 Lead-Tin Alloy | 8.5 |
| 20/80 Lead-Tin Alloy | 8.7 |
| Sn62 (62% Tin, 36% Lead) | 11.9 |
| Sn63 (63% Tin, 35% Lead) | 11.5 |
| Pure Tin | 13.9 |
| 80/20 Gold-Tin Alloy | 14.51 |
| 88% Gold, 12% Germanium | 14.67 |
| 82% Gold, 18% Indium | 14.9 |
- Lead-based solders, like 15/85 lead-tin alloy, have lower conductivity scores than some lead-free options.
- Solders with more lead, such as Sn62 and Sn63, show better conductivity, so you can use them in different electrical jobs.
- Lead-free solders can reach higher conductivity scores, which means they work well in electrical applications.
Safety Considerations
You must handle lead with care. Lead can be harmful if you breathe in dust or touch it often. Always wash your hands after working with lead. Many industries now use lead-free solders to keep you safe. Lead’s non-conductive properties help in shielding cables and batteries, but safety comes first. You should always follow safety rules when using lead in any project.
Titanium
Properties
Titanium is a strong and light metal. It looks silver-gray and does not rust fast. This metal stays safe in air and water. Most chemicals do not harm it. You can bend titanium and it will not snap. It melts only at very high heat, about 1,668°C (3,034°F). Titanium does not carry electricity as well as copper or aluminum. Its electrical conductivity is lower, so it slows electric current. This makes titanium different from metals used for wires. It acts more like non-conductive materials because electricity moves slower in it. Many people put titanium on lists of non-conductive metals for this reason.
Titanium also fights off corrosion. Saltwater, acids, and chlorine do not hurt it fast. You can use titanium in tough places where other metals would break. It is non-toxic, so doctors use it in medical tools and implants. Most people do not get allergies from titanium. It lasts a long time without falling apart.
Industrial Uses
Many industries use titanium for important jobs. Its special mix of features makes it useful in many ways. Here are some things about titanium in industry:
- Titanium’s moderate conductivity helps control heat and gives electrical insulation in hard places.
- You find titanium in airplane engines and spacecraft because it is strong and light.
- Power plants use titanium for pipes and heat exchangers. It does not rust, so it lasts longer.
- Medical companies use titanium for fake joints and dental implants. It is safe for the body and does not break down.
- Sports gear, like golf clubs and bikes, use titanium for strength without adding much weight.
Titanium’s electrical conductivity is one reason people pick it for these uses. Even though it is not the best at carrying electricity, its other features make it a great choice. You can use titanium when you need a metal that does not rust, stays strong, and handles heat well. Its non-conductive nature helps when you want to slow or block electric current. This makes titanium a smart choice for many new products.
Note: When you pick materials for electrical safety, remember that titanium acts differently than metals that carry electricity well. It works best where you need strength and safety, not just conductivity.
Uses And Applications
Electrical Safety
It is important to know about electrical safety. Non-conductive metals help protect you from shocks. These metals slow down electricity in panels and outlets. This gives you extra safety. You see them in fuse boxes and circuit breakers. They act as barriers so electricity stays where it should. You can trust these metals to keep places safer. Many safety devices use non-conductive metals to stop fires. They also help prevent accidents. Grounding is a big part of electrical safety. Grounding uses metals to send unwanted electricity into the ground. This keeps you safe if wires break or devices fail. Good grounding means you pick the right metal.
Grounding And Low Conductivity
You might ask what grounding does in a system. Grounding connects parts to the earth. This gives electricity a safe path. Using metals with low conductivity slows electricity down. This helps in special cases where you want to limit speed. You see this in sensitive equipment. Too much electricity can cause damage. Grounding stops shocks, lowers fire risk, and protects devices. Factories use grounding to keep workers and machines safe. Always check the metal you use for grounding. Some metals work better than others. Picking the right metal helps you avoid problems and keeps things safe.
Tip: Always follow safe grounding rules. Pick metals that fit your needs. Check conductivity before you use them.
Industrial Benefits
Industries use low-conductivity metals for many reasons. These metals help control heat and electricity in machines. Here is what happens when you use these metals:
- Low-conductivity metals are in laser cutters and precision tools. They keep machines stable and safe.
- Electronics makers use these metals to limit heat and add resistance. Nickel alloy resistors are one example.
- Aerospace engineers use these metals in systems that manage heat. This helps planes and rockets stay safe and work well.
- Medical devices, like MRI machines and defibrillators, use non-conductive metals. This makes them safer and more reliable.
- Some alloys can handle high heat. You find them in cars, airplanes, and big machines.
- Low thermal conductivity matters in heating elements and cryogenic supports. These parts need to control heat flow.
Low-conductivity metals are important in many industries. They help you build safer and longer-lasting products.
Misconceptions About Non-Conductive Metals
Common Myths
Some people think non-conductive metals stop all electricity. This is not true. Every metal lets some electricity pass, even if it is slow. You cannot use any metal for grounding. Some metals are better for grounding than others. You need to know which metals slow electricity and which ones let it move fast. This helps you stay safe when you build or fix things.
Many people believe non-conductive metals are always safer. That is not correct. If you pick the wrong metal for grounding, it can be dangerous. You must choose metals that fit the job. Always check each metal’s properties before using it for grounding.
Everyday Misunderstandings
You might hear wrong ideas about non-conductive metals at home or work. Some people think these metals are weak and cannot handle big jobs. Others believe insulation on metals wears out fast. Some say these metals are not good for heavy work. The table below shows what people think and what is true:
| Misconception | Reality |
|---|---|
| Non-conductive materials are weaker. | FRP scaffolds meet or exceed the strength standards required for construction safety. |
| FRP insulation fades over time. | The insulation is inherent, not a coating—it remains stable for decades. |
| It’s not suitable for heavy industrial work. | FRP scaffolding is used in oil, gas, and high-voltage sectors daily with proven reliability. |
Non-conductive materials like FRP are strong and last a long time. They work well in tough places. Do not judge a metal by how it looks or what people say. Always check facts before using something for grounding.
Note: Picking the right metal for grounding keeps you and your equipment safe. Do not trust myths or rumors.
Scientific Clarification
Science helps you learn what non-conductive metals really are. Some materials change how they act in different places. For example, hydrogen can become a metal and conduct electricity under very high pressure. This shows materials you think are non-conductive can act like metals in special conditions. You need to look at the environment and the job before choosing a metal for grounding. Science teaches you that context matters. You cannot say a metal is always non-conductive or always safe for grounding. You must test and check every time.
Conductivity changes with temperature, pressure, and other things. Always use the right tools to test metals before grounding. This helps you avoid mistakes and keeps your projects safe.
Identifying Non-Conductive Metals
Testing Methods
You can use several methods to find out what metals are non-conductive. In labs, scientists use special tools to test metals. Here are some common ways:
- The eddy-current method checks how electricity moves through a metal. This method works well for nonmagnetic metals with flat or slightly curved surfaces. You can use it to learn about the type of metal, how it was made, and if it has changed over time.
- The four-point probe method uses four small points to touch the metal. It measures how much resistance the metal has. If the resistance is high, the metal is less likely to carry electricity well. This method follows a standard called ASTM B 193-87.
- The standard test method (ASTM B 193-87) helps you find the resistivity of a metal. You can use this information to figure out if a metal is good or bad at letting electricity pass.
These tests help you know what metals to use for grounding. You want to pick metals that fit your needs and keep you safe.
Visual Clues
You can sometimes spot non-conductive metals by looking at them. Here are some things to watch for:
- Color: Some non-conductive metals, like bismuth, have a pinkish or silvery look. Lead looks dull gray. Titanium has a silver-gray shine.
- Weight: Non-conductive metals often feel heavier or lighter than you expect. Bismuth feels heavy for its size. Titanium feels light but strong.
- Surface: Some metals do not rust or change color in air or water. Titanium and bismuth stay shiny. Lead looks dull and soft.
- Markings: Some metals have labels or stamps that tell you what they are. Always check for these before using a metal for grounding.
You should remember that looks can fool you. Always test a metal if you are not sure. Visual clues help, but they do not replace real tests.
Safety And Grounding
You must think about safety when you work with metals. Grounding is a key part of electrical safety. You use grounding to send extra electricity safely into the earth. This keeps you safe from shocks and stops fires. When you pick a metal for grounding, you need to know if it is non-conductive or has low conductivity. Using the wrong metal for grounding can put you at risk.
Here is a quick checklist for grounding safety:
| Step | What to Do |
|---|---|
| Check the metal | Test or look for clues about the metal’s type. |
| Use the right metal | Pick metals that work well for grounding. |
| Inspect coatings | Make sure coatings do not block grounding. |
| Follow rules | Use grounding methods that meet safety codes. |
| Stay alert | Watch for signs of damage or wear. |
Tip: Always use the best metal for grounding. Do not guess. Testing and checking help you avoid mistakes.
You can keep yourself and others safe by knowing what metals to use for grounding. Good grounding protects your home, your tools, and your life.
Final Conclusion
You now know non-conductive metals have special features. Some, like Ba2Na1−OsO6, stay non-conductive because of their unique electronic structure. This means they cannot become conductors. Learning about conductivity helps you build safer electrical systems. It also helps stop dangers like shocks or fires. Engineers use this information to pick the best materials. They want energy to move well and keep things insulated. You can use this knowledge in many projects. These include cryogenic insulation, aerospace parts, electronic substrates, and medical devices. When you understand these metals, you can make better choices in materials science.
FAQ
What is a non-conductive metal?
A non-conductive metal does not let electricity move fast. These metals have high resistance. They slow down electric current more than copper or aluminum.
What makes a metal less conductive?
Metals with fewer free electrons conduct less electricity. If a metal has impurities or a messy atomic structure, electrons cannot move well. This makes the metal resist electric current.
What are some examples of non-conductive metals?
Bismuth, titanium, lead, and tungsten are examples. These metals do not carry electricity as well as copper or silver. They are good for jobs where you want to slow down electricity.
What industries use non-conductive metals?
Aerospace, medical devices, electronics, and power plants use these metals. They help control heat, add safety, and protect equipment from electrical problems.
What is the difference between non-conductive metals and insulators?
Non-conductive metals slow electricity but still let some current pass. Insulators, like rubber or glass, block electricity almost all the way. You use insulators when you need full protection.
What tests help you identify non-conductive metals?
You can use the eddy-current method or the four-point probe method. These tests check how much resistance a metal has. If resistance is high, the metal is less conductive.
What safety tips should you follow with non-conductive metals?
Always test the metal before using it for grounding. Look for coatings or damage. Follow safety codes and pick the right metal for each job. This keeps you and your equipment safe.