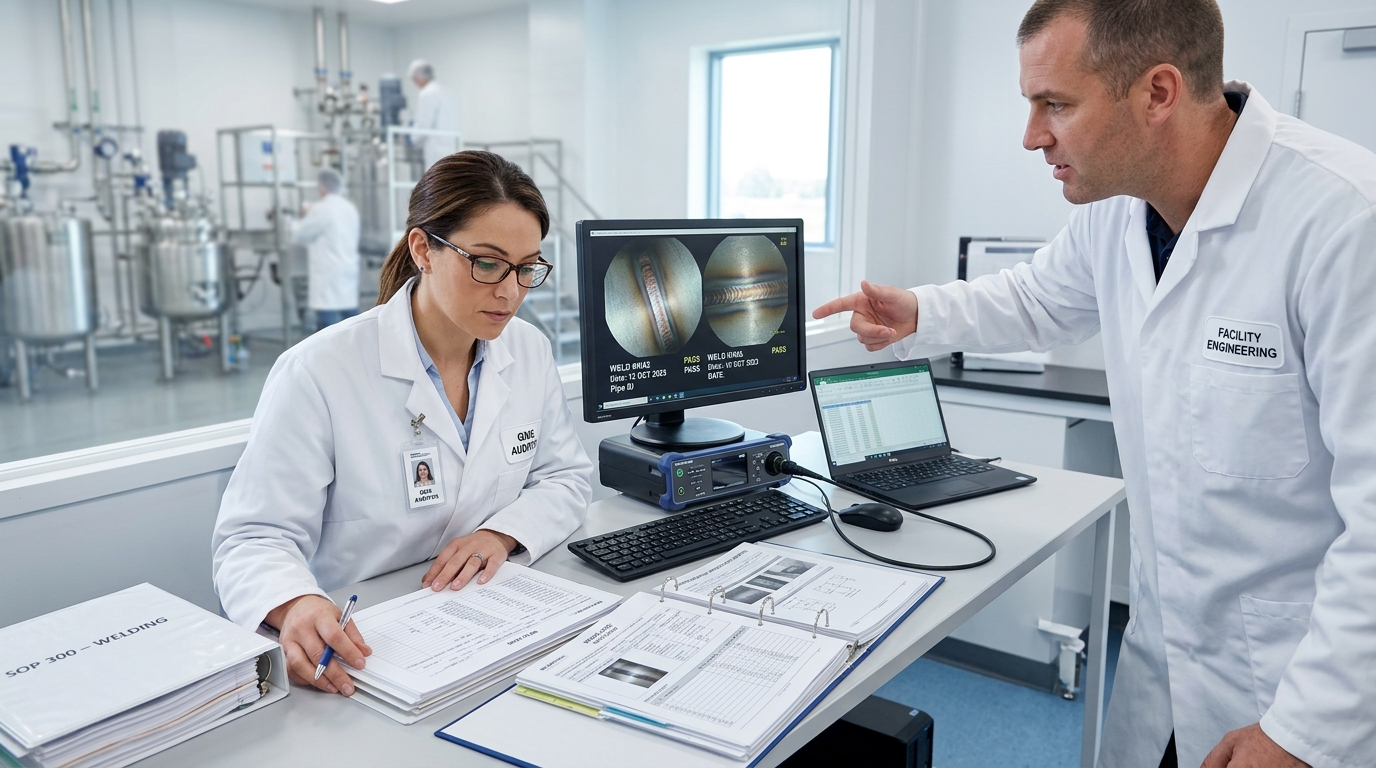
A single contaminated weld seam on a stainless steel process line can harbor enough biofilm to trigger a full batch rejection—costing a pharmaceutical manufacturer anywhere from $50,000 to over $500,000 per incident. Weld cleaning in the pharmaceutical industry refers to the post-weld removal of heat tint, oxidation, and surface contaminants from stainless steel joints to restore corrosion resistance, ensure hygienic surface finishes, and meet strict FDA and ASME BPE compliance standards. This guide breaks down the methods, specifications, and real-world workflows that keep pharma-grade piping systems safe for product contact.
What Is Weld Cleaning in the Pharmaceutical Industry and Why It Matters
Every weld leaves a mark. When stainless steel is welded in pharmaceutical manufacturing, the intense heat generates a cascade of surface defects — heat tint (that rainbow discoloration), oxide scale, and microscopic iron particles embedded in the metal’s surface. Weld cleaning is the process of removing these contaminants and restoring the stainless steel’s protective chromium oxide layer, which is what makes the material “stainless” in the first place.
In pharma, the stakes are different from structural fabrication. A discolored weld on a building beam is cosmetic. A discolored weld inside a WFI (Water for Injection) loop or a CIP-capable vessel is a contamination risk. Those heat tint oxides — typically ranging from light straw (about 300°C exposure) to dark blue or black (above 600°C) — create a rough, iron-rich surface where bacteria can harbor and corrosive attack can begin. Embedded free iron particles accelerate pitting corrosion, which creates microscopic crevices nearly impossible to sanitize.
Weld cleaning in the pharmaceutical industry specifically targets these post-weld defects on product-contact surfaces. The goal isn’t just visual cleanliness. It’s achieving a measurable surface condition — typically below 0.8 µm Ra for process piping and below 0.4 µm Ra for high-purity applications — that resists microbial adhesion and withstands repeated exposure to cleaning agents like sodium hydroxide and peracetic acid.
Patient safety is the bottom line. Residual weld contamination can leach metallic ions into drug products, promote biofilm formation, or cause batch failures during environmental monitoring. The ASME BPE standard exists precisely because generic fabrication tolerances aren’t sufficient for pharmaceutical-grade systems. Getting weld cleaning right protects both the product and the people who depend on it.
Heat tint and oxidation on a stainless steel pharmaceutical weld before weld cleaning
Why Weld Cleaning Is Critical in Pharmaceutical Processing Environments
Uncleaned welds don’t just look bad. They create invisible traps where bacteria colonize, corrosion starts, and product integrity falls apart. In pharmaceutical processing, a single compromised weld zone inside a vessel or transfer line can harbor Pseudomonas or Burkholderia cepacia biofilms that survive CIP cycles and resist chemical sanitizers. Once established, these biofilms shed organisms directly into process fluids—triggering batch failures, costly investigations, and potential FDA warning letters.
The heat-affected zone (HAZ) surrounding a weld is where most problems originate. Welding depletes the chromium oxide layer that gives stainless steel its corrosion resistance, leaving behind a rough, discolored surface with iron-rich inclusions. According to ASME BPE standards, these oxide layers must be removed and the passive film restored to maintain hygienic conditions. Without proper weld cleaning in pharmaceutical industry applications, pitting corrosion can initiate within weeks in high-purity water systems operating above 60°C.
The financial consequences are severe. A rejected batch of injectable product can represent $500,000 or more in lost material alone—before you factor in deviation investigations, revalidation, and production downtime. Rouge contamination traced back to poorly cleaned orbital welds in WFI distribution loops has forced complete system replacements at multiple facilities, with remediation costs exceeding $2 million per incident.
Cleanability is the core issue. Surface roughness at weld joints that exceeds specification creates microscopic crevices where residues accumulate and resist rinsing. Even a well-designed CIP program cannot compensate for a surface that physically traps soil. This makes thorough weld cleaning a prerequisite—not a finishing touch—for any system expected to meet sterility assurance requirements.
Uncleaned stainless steel weld in pharmaceutical piping showing heat tint and surface oxidation
Regulatory Standards and Compliance Requirements for Pharmaceutical Welds
Pharmaceutical welds sit at the intersection of multiple regulatory frameworks, and each one has teeth. The FDA’s 21 CFR Part 211 doesn’t mention welds explicitly, but its cGMP requirements for equipment surfaces are unambiguous: all contact surfaces must be nonreactive, nonadditive, and nonabsorptive. During facility inspections, FDA investigators interpret this to mean every weld on a product-contact system must be smooth, fully penetrated, and free of discoloration or oxidation that could harbor contamination.
ASME BPE (Bioprocessing Equipment) is where the specifics live. This standard defines acceptable weld bead geometry, concavity limits, and heat-affected zone discoloration levels on a scale from I to IX. Most pharmaceutical manufacturers target Level II or better for high-purity systems. Anything beyond Level V typically triggers rejection. The standard also specifies borescope inspection requirements — auditors expect photographic documentation of internal weld quality at every joint, not just spot checks.
ISPE Baseline Guides complement these standards by outlining commissioning and qualification protocols. They establish that weld cleaning in the pharmaceutical industry must be documented with traceable records linking each weld to its inspection results, welder ID, and cleaning method used. Miss one weld log, and an auditor can flag your entire installation package.
What catches companies off guard during audits is the documentation burden. Inspectors don’t just look at the welds themselves — they review your weld maps, coupon test results, and cleaning validation records as a complete package. A physically perfect weld without proper paperwork is, from a compliance standpoint, equivalent to a defective one. Proper weld cleaning in pharmaceutical industry settings is only half the battle; proving you did it correctly is the other half.
FDA inspector reviewing pharmaceutical weld documentation and borescope inspection images during a facility compliance audit
Surface Finish Requirements and Ra Value Specifications for Pharma-Grade Welds
Surface roughness isn’t cosmetic. It’s a measurable contamination risk. In pharmaceutical piping and vessels, the Ra value—arithmetic average roughness—determines how easily a surface can be cleaned, sanitized, and kept free of microbial harboring. A weld zone that exceeds the specified Ra creates microscopic peaks and valleys where product residue clings and biofilm takes root.
Ra Targets by Application
The required finish depends on what flows through the system. Water for Injection (WFI) systems typically demand Ra ≤ 0.5 µm (20 µin), with many facilities specifying Ra ≤ 0.38 µm (15 µin) on product-contact surfaces. Clean steam generators and purified water loops usually fall in the 0.5–0.8 µm range. API processing equipment and bioreactor vessels often sit between 0.4 and 0.6 µm, though some high-potency applications push tighter. The ASME BPE standard provides detailed surface finish classifications—SF1 through SF6—that map directly to these Ra thresholds and specify whether mechanical polish or electropolish is required.
Mechanical Polish vs. Electropolish
Mechanical polishing abrades the surface with progressively finer grits to reduce roughness. It works. But it leaves directional scratch patterns that can trap contaminants at the microscopic level. Electropolishing, by contrast, removes material electrochemically, preferentially dissolving peaks to produce a smoother, more passive surface. An electropolished finish at Ra 0.5 µm is functionally cleaner than a mechanically polished surface at the same Ra reading because the surface profile is rounder, with fewer sharp asperities.
Matching the Weld Zone to Parent Material
Here’s where weld cleaning in the pharmaceutical industry gets demanding. The weld and heat-affected zone must meet the same Ra specification as the surrounding tube or vessel wall. A beautifully electropolished pipe with a rough weld bead at Ra 1.2 µm defeats the purpose entirely. Post-weld finishing—whether by electrochemical cleaning, localized electropolishing, or controlled mechanical blending—must bring that zone into spec. Surface profilometry using contact stylus instruments (per ISO 4287) or optical profilometers verifies the result. Measurements are typically taken at three or more points across the weld centerline and HAZ, and all readings must fall within the specified tolerance before the joint is accepted.
Surface profilometry measurement on pharmaceutical-grade stainless steel weld for Ra value verification
Electrochemical vs. Chemical vs. Mechanical Weld Cleaning Methods Compared
Three methods dominate. Each carries trade-offs that matter when the finished surface must meet pharma-grade specifications.
| Criteria | Electrochemical | Chemical (Pickling Paste) | Mechanical (Grinding/Blending) |
|---|---|---|---|
| Heat Tint Removal | Excellent — removes discoloration without material loss | Good — dissolves oxide layer chemically | Variable — depends on abrasive grit and operator skill |
| Surface Finish Preservation | Maintains original Ra; typically adds <0.05 µm variance | Can etch aggressively if left too long, raising Ra by 0.1–0.3 µm | Often degrades finish; 80-grit flap discs can push Ra above 0.8 µm |
| Speed (per linear meter) | 3–6 minutes | 20–60 minutes dwell time | 5–15 minutes, plus re-polishing |
| Safety / Hazards | Low — uses phosphoric acid electrolyte at mild concentrations | High — contains hydrofluoric and nitric acid mixtures | Moderate — airborne particulate, noise exposure |
| Environmental Impact | Minimal waste; electrolyte is recyclable | Generates hazardous acid waste requiring neutralization | Abrasive waste; risk of iron contamination from non-dedicated tools |
| Best Suited Grades | 304L, 316L, duplex — handles all common pharma alloys | Effective on 304L/316L; aggressive on thinner gauges | Acceptable for structural welds, risky on product-contact surfaces |
Mechanical methods remain common for rough structural joints, but they introduce embedded abrasive particles and directional scratch patterns that trap biofilm. For product-contact welds, the ASME BPE standard effectively rules out aggressive grinding on surfaces requiring Ra ≤ 0.5 µm. Chemical pickling works, yet the hydrofluoric acid component creates serious handling and disposal burdens — a real concern in cleanroom-adjacent environments where fume migration must be controlled.
Electrochemical weld cleaning in pharmaceutical industry applications has gained ground precisely because it threads the needle: fast oxide removal, simultaneous passivation, and negligible impact on the base metal’s finish. The equipment is portable, the electrolyte is far less hazardous than pickling paste, and operators need less training to produce consistent results across complex joint geometries like tee fittings and reducer transitions.
No single method suits every scenario. But when the priority is preserving a validated surface finish while meeting tight turnaround schedules, electrochemical cleaning outperforms the alternatives on nearly every metric that matters to pharma fabricators.
How Electrochemical Weld Cleaning Works and When to Use It in Pharma Applications
The process is deceptively simple. A carbon fiber brush soaked in phosphoric acid-based electrolyte is connected to a power unit that delivers low-voltage AC or DC current—typically between 6 and 40 volts. When the brush contacts the weld surface, the electrical current drives an electrochemical reaction that dissolves the heat tint oxide layer while simultaneously forming a fresh, chromium-rich passive film on the stainless steel beneath.
What makes this particularly relevant for weld cleaning in pharmaceutical industry applications is the dual action: cleaning and passivation happen in a single pass. The electrolyte chemistry matters enormously here. Most professional-grade solutions use a blend of phosphoric acid at 15–25% concentration with proprietary surfactants that control the reaction rate and prevent pitting. Cheaper formulations sometimes substitute with sulfuric acid blends, but these carry a higher risk of intergranular attack on 316L stainless—a real problem when your target Ra is 0.5 µm or below.
Operating parameters need tight control. Current density typically runs between 0.5 and 2.0 A/cm², and brush speed should stay consistent at roughly 3–5 cm per second. Too slow, and you risk localized overheating. Too fast, and the oxide layer won’t fully dissolve. According to guidance from the ASME BPE standard, any cleaning method used on bioprocessing surfaces must leave a verifiably passive, corrosion-resistant finish—electrochemical cleaning meets this threshold when executed correctly.
When should you choose this method in pharma fabrication? Orbital welds on small-bore tubing (6–50 mm OD) are the sweet spot. It’s also the go-to for field repairs where chemical immersion isn’t feasible and mechanical abrasion would compromise surface finish. The equipment is portable, the process generates minimal waste, and total material removal stays under 2 µm—far less than any grinding or blending operation could achieve.
Passivation After Weld Cleaning and How to Verify Results
Cleaning the weld is half the job. The other half is rebuilding what welding destroyed: the chromium oxide layer that gives stainless steel its corrosion resistance. This passive layer—typically only 1–5 nanometers thick—gets disrupted by heat, and without deliberate restoration, even a perfectly cleaned weld will corrode in aggressive pharmaceutical process fluids.
Citric Acid vs. Nitric Acid Passivation
Two chemistries dominate. Nitric acid (20–50% concentration at 20–50°C) has been the industry standard for decades, and it works. But it’s hazardous to handle, generates toxic fumes, and creates disposal headaches. Citric acid passivation (4–10% concentration) achieves comparable chromium-to-iron ratios on the surface while being far safer for operators and the environment. ASTM A967/A967M recognizes both chemistries and specifies acceptable procedures for each, including immersion, spray, circulation, and gel application methods.
For weld cleaning in pharmaceutical industry applications, citric acid has gained significant ground. Many facilities now specify it as the default, reserving nitric acid for stubborn cases or legacy SOPs that haven’t been updated.
Verification Techniques That Actually Prove Passivation Worked
ASTM A967 and ASTM A380 outline several acceptance tests. The ferroxyl test (copper sulfate solution applied to the surface) detects free iron—a blue reaction means the surface failed. Quick, cheap, and effective for field use. Water break testing is even simpler: a uniform water sheet across the surface indicates a clean, passive layer, while beading or breaking suggests contamination.
For the highest assurance, XPS (X-ray Photoelectron Spectroscopy) provides a quantitative chromium-to-iron ratio. A Cr/Fe ratio above 1.5:1 generally indicates robust passivation. XPS requires lab equipment and isn’t practical for every weld, but it’s the gold standard when qualifying a new process or resolving disputes about surface quality.
Best Practices and Step-by-Step Workflow for Pharmaceutical Weld Cleaning
A repeatable workflow eliminates guesswork. It also eliminates audit findings. Here’s a practical sequence that covers pre-weld preparation through final sign-off, built around the reality that most rework stems from skipped steps rather than bad technique.
Pre-Weld Preparation
Start with material verification and joint fit-up. Gaps wider than 0.5 mm invite incomplete fusion and create crevices that no amount of post-weld cleaning can fully resolve. Use argon (99.996% purity minimum) as your back-purge gas, and confirm oxygen levels drop below 50 ppm before striking the arc—many shops target under 25 ppm for high-purity systems. Wipe the joint zone with isopropyl alcohol and lint-free cloths to remove oils, marker residue, and shop dust.
Post-Weld Cleaning Sequence
- Visual inspection first. Reject any weld showing sugaring, porosity, or undercut before cleaning begins—polishing a defective weld wastes time and hides problems.
- Electrochemical cleaning pass. Run the brush at the manufacturer’s recommended amperage, maintaining steady contact for 1–2 seconds per centimeter of weld length.
- Neutralize and rinse. Apply neutralizing fluid immediately, then rinse with deionized water (conductivity below 1.0 µS/cm).
- Passivation verification. Confirm chromium oxide layer integrity using the methods covered in the previous chapter.
- Surface roughness check. Measure Ra at three points along the weld; all readings must fall within the specification defined on the isometric drawing.
Common Mistakes That Trigger Rework
Skipping the oxygen purge check is the single biggest source of weld discoloration in pharmaceutical piping. Another frequent issue: operators reuse contaminated cleaning fluid, which deposits iron particles back onto the surface. The ASME BPE standard specifically addresses cleanliness of contact materials for this reason. Document every step with photos and readings—effective weld cleaning in the pharmaceutical industry depends as much on traceability as on technique.
Frequently Asked Questions About Weld Cleaning in Pharma Manufacturing
Is some weld discoloration acceptable?
It depends on the zone classification. Light straw coloring (AWS D18.2 color #1–#3) on non-product-contact surfaces may pass inspection in utility systems. But any visible heat tint on product-contact welds in high-purity water or API processing lines is typically rejected. The discoloration indicates a depleted chromium oxide layer, which means compromised corrosion resistance regardless of how faint it looks.
Does orbital welding eliminate the need for post-weld cleaning?
No. Orbital welding with proper purge gas produces far cleaner welds—often with zero discoloration—but it doesn’t guarantee a fully passivated surface. The heat-affected zone still experiences chromium depletion at the microstructural level. Most specifications still require passivation verification via ferroxyl or water contact angle testing, even on pristine-looking orbital welds.
How often should installed systems be repassivated?
There’s no universal interval. Facilities running aggressive CIP cycles with peracetic acid or high-concentration sodium hydroxide may need repassivation every 12–18 months. Systems handling WFI with minimal chemical exposure can go 3–5 years. The real trigger should be rouge formation or declining ASTM A967 test results, not a calendar date.
Is laser weld cleaning viable for pharmaceutical applications?
Laser ablation removes oxides effectively and leaves no chemical residue. The technology works well for accessible, flat surfaces. The limitation? Most pharma piping involves small-bore tubing and tight internal geometries where a laser head simply can’t reach. For external welds on vessels and large-diameter spools, it’s a legitimate option. For 1″–3″ process tubing, electrochemical methods remain more practical.
Can you clean welds on already-installed systems without dismantling them?
Yes, but access dictates the method. Electrochemical weld cleaning in pharmaceutical industry applications often uses flexible brush extensions that reach welds inside installed pipe runs through open tri-clamp connections. For internal surfaces beyond brush reach, circulating chemical passivation solutions through the system is the fallback. Full disassembly is only necessary when physical inspection or borescope verification is required by the quality protocol.
Key Takeaways for Implementing Effective Pharmaceutical Weld Cleaning Programs
Getting weld cleaning in the pharmaceutical industry right comes down to three pillars: choosing the correct method, documenting every step, and verifying results with objective measurements. Skip any one of these, and your program has a gap an auditor will find.
Electrochemical cleaning handles the majority of pharma-grade applications because it cleans, passivates, and preserves surface finish in a single operation—but it’s not universal. Thick oxide scale from multi-pass welds on heavy vessels may still require mechanical prep before electrochemical finishing. Match the method to the weld, not the other way around.
Quick-Reference Checklist for Your Weld Cleaning Program
- Method selection: Confirm the cleaning approach achieves ≤ 0.5 µm Ra (or your spec’s target) without embedding contaminants. Validate on sample coupons before production use.
- Passivation verification: Require ferroxyl testing or ASTM A967 water immersion on every cleaned weld. Record pass/fail with photo evidence.
- Operator qualification: Train and certify each technician. Re-certify annually, and after any process change.
- Documentation package: Each weld needs a traceable record—weld ID, cleaning method, operator, date, passivation test result, and Ra measurement where specified.
- Chemical control: Log electrolyte or pickling solution lot numbers. Confirm rinse water meets FDA purified water conductivity limits (≤ 1.3 µS/cm at 25 °C).
- Audit readiness: Store records for a minimum of 3 years—longer if your quality agreement demands it. Organize by system or work order so retrieval takes minutes, not days.
A solid program isn’t complicated. It’s consistent. Build the SOPs once, enforce them every time, and treat documentation as a deliverable equal to the weld itself. That discipline is what separates facilities that breeze through FDA inspections from those that scramble.