The melting point of stainless steel falls between roughly 1,375 °C and 1,530 °C (2,500–2,790 °F), depending on the specific grade and its alloy composition. That range matters more than a single number — grade 304 starts to melt around 1,400 °C, while grade 410 holds solid up to approximately 1,530 °C, a gap of 130 °C that can make or break a high-temperature design. This guide breaks down the exact melting ranges for every major stainless steel family, explains why alloying elements shift those numbers, and clarifies the practical differences engineers and fabricators actually need to care about.
What Is the Melting Point of Stainless Steel
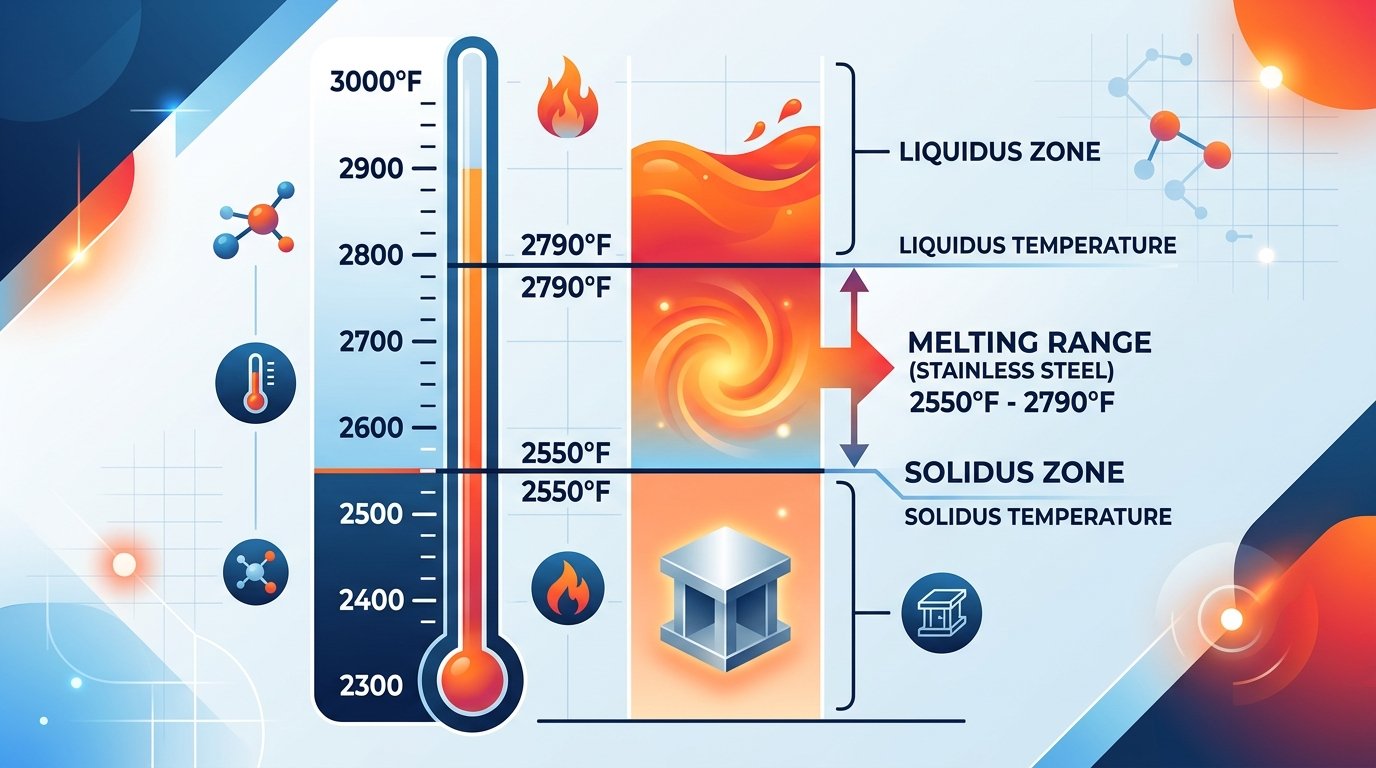
The melting point of stainless steel falls between 2,550°F and 2,790°F (1,400°C to 1,530°C), depending on the specific grade. That’s not a single temperature — it’s a range. And the distinction matters more than most people realize.
Stainless steel is an alloy, not a pure element. Pure iron melts at a clean 2,800°F (1,538°C). But the moment you add chromium, nickel, molybdenum, carbon, and other elements to create stainless steel, you shift the melting behavior. Each alloying element interacts with iron’s crystal structure differently, pulling the melting threshold up or down. Grade 304, for example, starts to melt around 2,550°F (1,399°C), while grade 430 holds out until roughly 2,600–2,750°F (1,425–1,510°C). That 200°F gap between grades can be the difference between a component surviving a furnace environment and one that doesn’t.
Why does this variation exist? Two reasons. First, different grades contain dramatically different chemical compositions — grade 316 packs 2–3% molybdenum that grade 304 lacks entirely. Second, stainless steel doesn’t transition from solid to liquid at one precise temperature the way ice does at 32°F. Instead, it passes through a “mushy zone” where solid and liquid phases coexist. Metallurgists refer to the start of melting as the solidus temperature and the point where everything is fully liquid as the liquidus temperature. The numbers you see quoted are typically this solidus-to-liquidus range, as documented by sources like the ASM International materials database.
Knowing the melting point of stainless steel by grade isn’t just academic trivia. It directly influences welding parameters, casting procedures, and high-temperature service limits — topics we’ll break down grade by grade in the sections below.
Stainless steel melting point range infographic showing solidus and liquidus temperatures from 2550 to 2790 degrees Fahrenheit
Why Stainless Steel Has a Melting Range Instead of a Single Fixed Point
Pure iron melts at exactly 2,800°F (1,538°C). Pure nickel, 2,651°F (1,455°C). Pure chromium, 3,465°F (1,907°C). Each of these elements has a single, sharp melting point — one precise temperature where solid becomes liquid. Stainless steel doesn’t work that way.
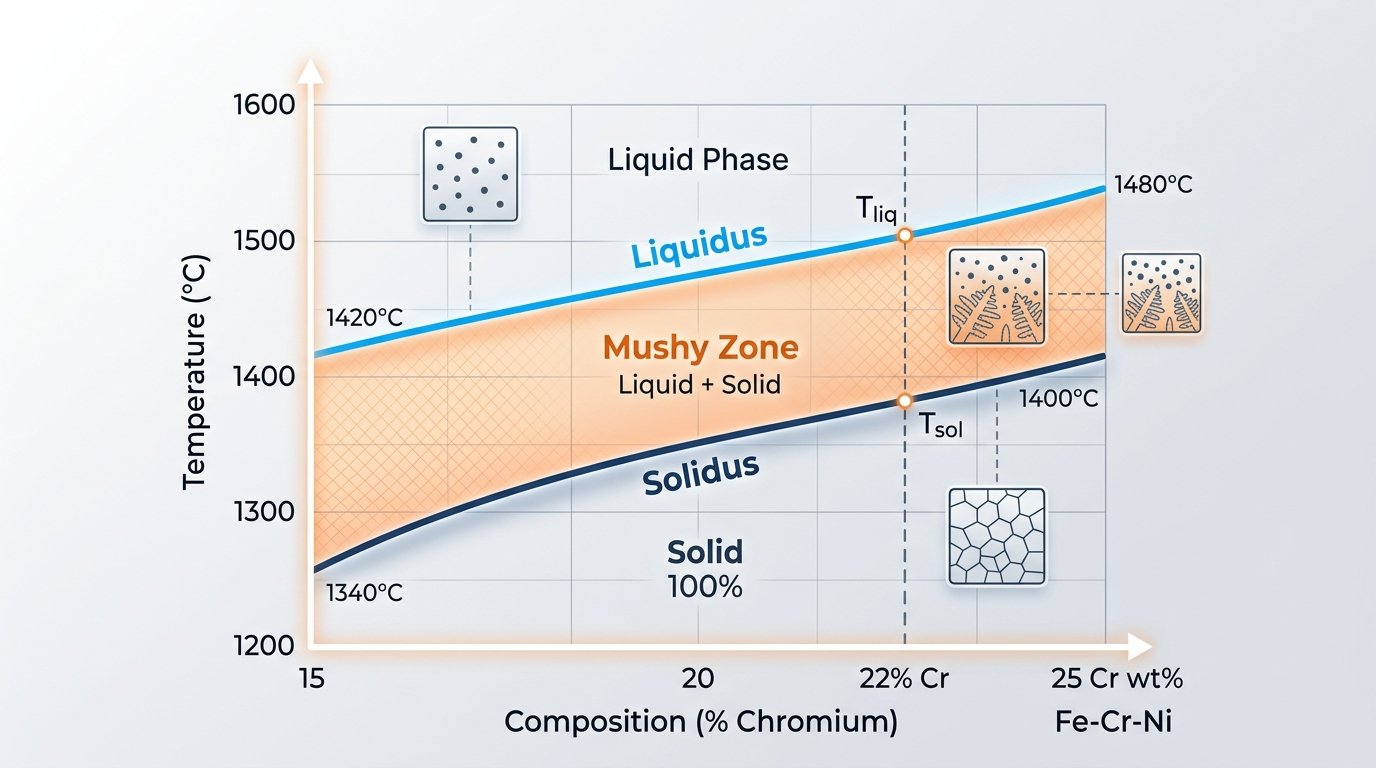
The reason comes down to basic metallurgy. Stainless steel is an alloy — a deliberate mixture of iron with chromium (at least 10.5%), plus varying amounts of nickel, molybdenum, manganese, carbon, and other elements. When you heat a multi-element alloy, the different atomic species in the crystal lattice don’t all lose their bonding energy at the same temperature. Instead, certain regions of the microstructure — particularly grain boundaries and areas enriched in lower-melting-point elements — begin to liquefy first. This creates a two-phase “mushy zone” where solid and liquid coexist.
Metallurgists define this behavior using two boundaries. The solidus is the temperature where the first liquid appears. The liquidus is where the last solid crystal dissolves. Between these two temperatures, the alloy exists as a slushy mixture. For most stainless steel grades, this solidus-to-liquidus gap spans roughly 50°F to 150°F (30°C to 85°C), depending on compositional complexity.
This is why discussing the melting point of stainless steel always requires specifying a range rather than a single number. A grade like 316, with its added molybdenum, has a different phase diagram than 430, which contains almost no nickel. More alloying elements generally mean a wider melting range and a lower solidus temperature. The ASM International phase diagram database documents these transitions in detail for hundreds of alloy compositions.
Phase diagram illustrating the solidus-to-liquidus melting range of stainless steel alloy
Melting Points of Common Stainless Steel Grades — Complete Comparison Table
The table below consolidates solidus and liquidus temperatures for the most commonly specified grades. Solidus marks where melting begins; liquidus marks where the alloy is fully liquid. Every grade-specific value here aligns with data published by AK Steel (now Cleveland-Cliffs) product data sheets and ASM International references.
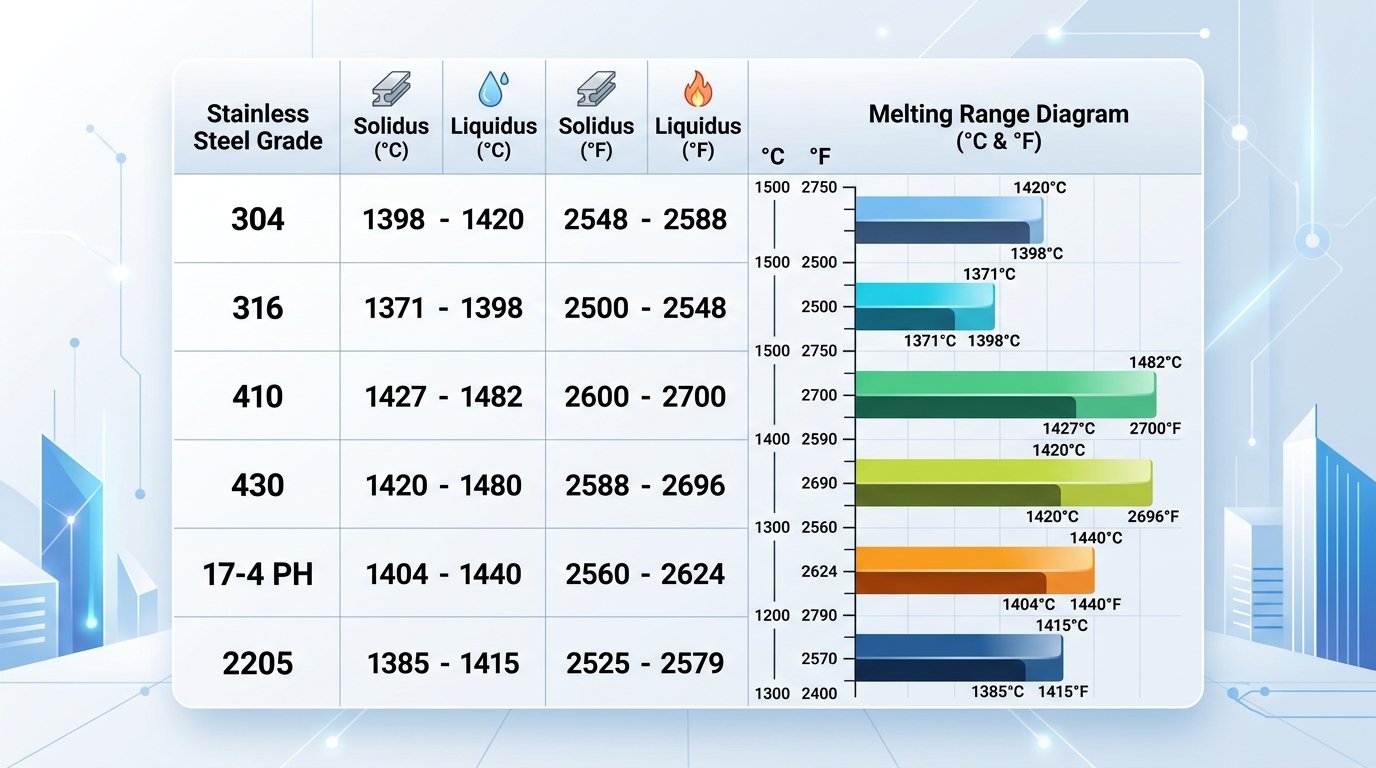
| Grade | Family | Solidus °F / °C | Liquidus °F / °C |
|---|---|---|---|
| 304 | Austenitic | 2,550°F / 1,399°C | 2,650°F / 1,454°C |
| 304L | Austenitic | 2,550°F / 1,399°C | 2,650°F / 1,454°C |
| 316 | Austenitic | 2,500°F / 1,371°C | 2,550°F / 1,399°C |
| 316L | Austenitic | 2,500°F / 1,371°C | 2,550°F / 1,399°C |
| 321 | Austenitic | 2,550°F / 1,399°C | 2,590°F / 1,421°C |
| 347 | Austenitic | 2,550°F / 1,399°C | 2,635°F / 1,446°C |
| 410 | Martensitic | 2,700°F / 1,482°C | 2,790°F / 1,532°C |
| 420 | Martensitic | 2,650°F / 1,454°C | 2,750°F / 1,510°C |
| 430 | Ferritic | 2,600°F / 1,427°C | 2,750°F / 1,510°C |
| 17-4 PH | Precipitation-Hardened | 2,560°F / 1,404°C | 2,625°F / 1,441°C |
| 2205 | Duplex | 2,550°F / 1,399°C | 2,640°F / 1,449°C |
A few patterns jump out immediately. The martensitic grades — 410 and 420 — sit at the top of the range, largely because they carry less nickel and molybdenum, elements that pull the melting point of stainless steel downward. Grade 316 and 316L land at the bottom, a direct consequence of their higher molybdenum content (2–3%).
Notice that the “L” variants (304L, 316L) share nearly identical melting ranges with their standard counterparts. The reduced carbon — typically capped at 0.03% — is too small a compositional shift to meaningfully alter thermal behavior. Where it matters is sensitization resistance during welding, not melting temperature.
Keep this table bookmarked. The chapters ahead break down each family in detail, explaining why these numbers differ and what they mean for real fabrication decisions.
Melting point comparison table for common stainless steel grades showing solidus and liquidus temperatures
Austenitic Grades — 304, 316, 321, and 347
Austenitic stainless steels dominate global consumption, accounting for roughly 70% of all stainless steel produced. Their face-centered cubic crystal structure, stabilized by nickel contents typically between 8% and 12%, gives them excellent ductility and corrosion resistance — but it also places their melting ranges in a fairly tight band compared to other families.
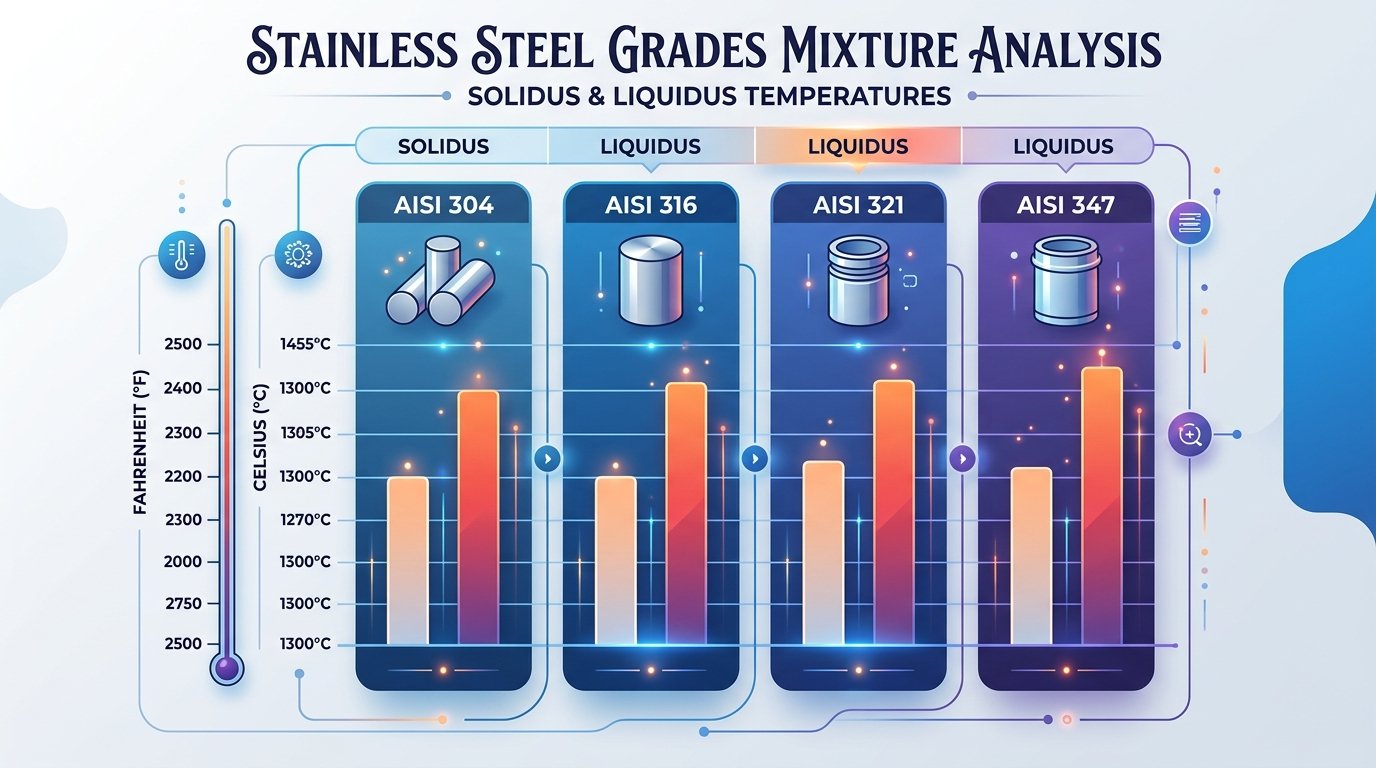
Grade 304, the workhorse of the group, carries about 18% chromium and 8–10.5% nickel. Its solidus sits near 2,550°F (1,399°C) and its liquidus around 2,650°F (1,454°C). That 100°F mushy zone matters during welding because the partially molten metal is vulnerable to hot cracking if heat input isn’t controlled.
Grade 316 adds 2–3% molybdenum to the mix. Molybdenum is a heavier element with a melting point of 4,753°F (2,623°C) on its own, yet in solid solution it actually depresses the alloy’s solidus slightly — down to roughly 2,500°F (1,371°C) — while pushing the liquidus to about 2,550°F (1,399°C). The melting point of stainless steel in the 316 family therefore starts lower than 304’s, a detail that catches some engineers off guard. According to data published by AK Steel (now Cleveland-Cliffs), this shift traces directly to how molybdenum disrupts the iron-chromium-nickel lattice.
Grades 321 and 347 are stabilized variants — 321 with titanium, 347 with niobium (columbium). These additions are small, usually under 1%, so they barely nudge the melting range. Both grades melt between roughly 2,550°F and 2,635°F (1,399–1,446°C), nearly identical to 304. Their real advantage is resistance to sensitization during prolonged high-temperature exposure, not a meaningful change in melting behavior.
One practical takeaway: when comparing the melting point of stainless steel across these four austenitic grades, the spread is only about 50°F at the solidus end. Composition tweaks matter far more for corrosion performance and weldability than they do for the temperature at which the metal actually turns liquid.
Melting range comparison chart for austenitic stainless steel grades 304, 316, 321, and 347
Ferritic and Martensitic Grades — 410, 420, and 430
Ferritic and martensitic stainless steels share a body-centered cubic (BCC) crystal structure at room temperature, which sets them apart from the face-centered cubic (FCC) austenitic family. They also contain little to no nickel — typically under 1% — and rely almost entirely on chromium for corrosion resistance. This compositional shift has a direct effect on the melting point of stainless steel in these grades, pushing solidus temperatures slightly higher than their austenitic counterparts.
Grade 430 — The Ferritic Baseline
Grade 430 is the most widely used ferritic stainless steel. With 16–18% chromium, no intentional nickel addition, and carbon held below 0.12%, it melts between 2,600°F and 2,750°F (1,425°C–1,510°C). The absence of nickel — a known melting-point depressant — is the primary reason its solidus sits about 50°F above 304. Ferritic grades are magnetic, less expensive, and commonly found in automotive trim, kitchen sinks, and architectural panels.
Grades 410 and 420 — Martensitic and Hardenable
Martensitic grades trade some corrosion resistance for hardness. Grade 410 contains 11.5–13.5% chromium with up to 0.15% carbon, and its melting range spans roughly 2,700°F to 2,790°F (1,480°C–1,530°C). Grade 420 bumps carbon to 0.15–0.40%, which narrows the solidus-liquidus gap slightly and lowers the solidus by about 20–30°F compared to 410. Higher carbon forms more chromium carbides during solidification, which concentrates solute in the remaining liquid and depresses the temperature at which the last solid forms. According to data published by AZoM, grade 420 typically begins melting near 2,650°F (1,450°C).
One practical takeaway: because martensitic grades harden through heat treatment rather than cold working, their thermal processing windows sit well below the solidus — usually 1,750°F to 1,850°F for austenitizing. Confusing service-temperature limits with actual melting behavior is a common engineering mistake covered later in this article.
Duplex and Precipitation-Hardened Grades — 2205 and 17-4 PH
Duplex and precipitation-hardened (PH) stainless steels occupy a different engineering niche than the austenitic and ferritic families covered earlier. Their chemistries are more complex, and that complexity shows up directly in their melting behavior.
Duplex 2205
Grade 2205 maintains a roughly 50/50 balance of austenite and ferrite phases. Its composition — about 22% chromium, 5% nickel, 3% molybdenum, and 0.17% nitrogen — gives it a solidus near 2,560°F (1,404°C) and a liquidus around 2,680°F (1,471°C). That 120°F melting range is tighter than most austenitic grades, partly because the high chromium and moderate nickel content keep the phase diagram relatively constrained. Nitrogen, often overlooked, acts as a powerful austenite stabilizer here without dramatically depressing the solidus the way carbon does.
17-4 PH
Precipitation-hardened 17-4 PH takes a fundamentally different approach. It achieves high strength — up to 200 ksi tensile in the H900 condition — through copper and niobium precipitates that form during aging heat treatments at 900°F to 1,150°F. The base chemistry (roughly 17% Cr, 4% Ni, 4% Cu, 0.3% Nb) produces a solidus of approximately 2,560°F (1,404°C) and a liquidus of 2,625°F (1,441°C). Copper is the key variable. It melts at just 1,984°F (1,085°C) on its own, and even at 4% concentration it measurably pulls the melting point of stainless steel downward compared to a straight martensitic grade like 410.
Both grades demand careful thermal control during fabrication. Duplex 2205 is sensitive to intermetallic sigma phase formation between 1,200°F and 1,750°F, while 17-4 PH can lose its precipitation-hardened strength if exposed to temperatures above its aging range for too long. Understanding these melting ranges matters because welding heat-affected zones can momentarily push local temperatures close to the solidus, risking incipient melting and hot cracking. The Nickel Institute’s guide to duplex stainless steels provides detailed phase stability data for engineers working with these alloys.
How Alloying Elements Like Chromium, Nickel, and Molybdenum Affect Melting Temperature
Each element dissolved in stainless steel nudges the solidus and liquidus lines in a specific direction. Understanding these individual contributions explains why the melting point of stainless steel shifts so dramatically from one grade to another — sometimes by more than 100°F.
Chromium — The Upward Push
Chromium melts at 3,465°F (1,907°C), well above iron’s 2,800°F. When you add 10.5–30% chromium to an iron matrix, it raises the liquidus temperature. Ferritic grades like 430, loaded with ~17% chromium and very little nickel, sit at the higher end of the stainless melting spectrum. More chromium also stabilizes the BCC ferrite phase, which inherently resists melting longer than austenite.
Nickel — The Downward Pull
Nickel’s own melting point is 2,651°F (1,455°C) — roughly 150°F below iron. Adding 8–14% nickel to austenitic grades depresses the solidus noticeably. That’s a big reason 304 (8% Ni) starts melting around 2,550°F while 430 (essentially 0% Ni) holds until 2,600°F. Higher-nickel superaustenitic alloys drop even further. The iron-nickel binary phase diagram from ASM International shows a continuous solidus depression as nickel content climbs.
Molybdenum, Carbon, and Manganese
Molybdenum melts at a scorching 4,753°F (2,623°C), yet its effect on stainless steel’s melting range is modest at typical 2–3% additions. It slightly widens the gap between solidus and liquidus rather than dramatically raising either. Carbon is the real wildcard — even 0.08% versus 0.03% can lower the solidus by 20–30°F because carbon is a potent freezing-point depressant in iron alloys. Manganese, usually present at 1–2%, behaves similarly to nickel: it stabilizes austenite and gently reduces the melting range.
The practical takeaway? Grades heavy in chromium and light in nickel melt higher. Grades rich in nickel, carbon, or manganese melt lower. Molybdenum’s contribution is secondary at the concentrations found in standard stainless steels.
Melting Point vs. Maximum Service Temperature — A Critical Distinction for Engineers
Knowing the melting point of stainless steel tells you when the alloy turns liquid. It does not tell you when it stops being useful. That gap is enormous — often 500°F to 1,000°F or more — and confusing the two numbers has led to catastrophic equipment failures in furnaces, exhaust systems, and chemical reactors.
Three mechanisms force engineers to derate stainless steel far below its melting range. Creep is the first: sustained stress at elevated temperature causes the metal to slowly deform, even when the applied load sits well under the room-temperature yield strength. For 304 stainless, creep becomes a serious design factor above roughly 1,000°F (538°C). Second, oxidation scaling accelerates as temperature climbs. Grade 304 forms a protective chromium-oxide layer up to about 1,650°F (900°C) in continuous service, but cyclic heating and cooling can spall that layer off at even lower temperatures, exposing fresh metal to rapid attack. Third, strength degradation is steep: 316L retains only about 60% of its room-temperature tensile strength at 1,000°F, according to data published by the Nickel Institute.
Here’s how maximum continuous service temperatures compare across popular grades:
| Grade | Melting Range (°F) | Max Continuous Service Temp (°F) |
|---|---|---|
| 304 | 2,550–2,650 | 1,650 |
| 316 | 2,500–2,550 | 1,700 |
| 321 | 2,550–2,600 | 1,650 |
| 410 | 2,700–2,790 | 1,200 |
| 2205 Duplex | 2,550–2,600 | 600 |
Notice something counterintuitive: duplex 2205 has a higher melting range than 316, yet its maximum service temperature is far lower. Above about 600°F (315°C), the sigma phase precipitates and embrittles the alloy. Martensitic 410 faces a different ceiling — its tempered hardness collapses if you exceed the original tempering temperature. The melting point of stainless steel is a thermodynamic property; the service limit is an engineering judgment that folds in creep life, corrosion kinetics, and microstructural stability. Design to the second number, not the first.
How Melting Point Impacts Welding, Casting, and Heat Treatment of Stainless Steel
The melting point of stainless steel isn’t just a material property on a datasheet — it directly governs how fabricators set up their processes. During welding, the solidus-to-liquidus gap determines weld pool behavior. A grade like 304, with a roughly 50°F (28°C) melting range, transitions from solid to liquid quickly, producing a fluid, fast-moving weld pool. Duplex 2205, with a wider mushy zone, tends to be sluggish and less forgiving of poor technique.
Casting fluidity follows a similar logic. Alloys with lower liquidus temperatures and narrower freezing ranges fill mold cavities more completely before solidification begins. That’s why foundries favor certain austenitic compositions for thin-walled castings — the metal stays liquid long enough to reach tight geometries. Wider freezing ranges increase the risk of centerline shrinkage porosity because the mushy zone traps pockets of liquid between growing dendrites.
Heat treatment introduces an even sharper concern: incipient melting. If a solution-annealing furnace overshoots and pushes a 17-4 PH part above its 2,560°F solidus, grain boundaries begin to liquefy while the bulk material stays solid. The result is irreversible cracking that no amount of post-weld heat treatment can fix. According to ASM International’s heat treatment guidelines, maintaining at least a 50°F margin below the solidus is standard practice for precipitation-hardened grades.
In the heat-affected zone (HAZ) during welding, peak temperatures can spike within a few millimeters of the fusion line. Ferritic grades like 430, with a solidus around 2,600°F, are particularly vulnerable to grain coarsening in this region. Fabricators compensate by limiting heat input — typically below 1.0 kJ/mm — to keep the HAZ narrow and minimize property degradation.
Frequently Asked Questions About Stainless Steel Melting Points
Does stainless steel melt at a higher temperature than carbon steel?
Not necessarily. Plain carbon steel (like AISI 1020) has a solidus around 2,760°F (1,515°C), which actually sits within — or even above — the melting range of many stainless grades. Grade 316, for instance, starts melting at 2,500°F (1,371°C), a full 260°F lower than 1020 carbon steel. The added nickel and molybdenum in austenitic stainless steels depress the solidus. So the assumption that “stainless = higher melting point” is flat-out wrong in many cases.
Which stainless steel grade has the highest melting point?
Among commonly available grades, ferritic 430 stainless reaches a liquidus of roughly 2,790°F (1,530°C), putting it near the top. Martensitic 410 is close behind at about 2,785°F. Both contain minimal nickel, which keeps their solidus and liquidus elevated. Specialty high-chromium ferritics like 446 (27% Cr) can push even slightly higher, though they see limited commercial use.
Can you melt stainless steel at home?
Technically, yes — but practically, it’s extremely difficult and dangerous. A propane forge tops out around 2,300°F, well short of the melting point of stainless steel. You’d need an oxy-acetylene setup (reaching ~5,600°F) or an electric arc furnace. Backyard foundry hobbyists on forums like IForgeIron occasionally report success with small induction furnaces, but the fumes from chromium and nickel are toxic. Proper ventilation and respiratory protection aren’t optional — they’re mandatory.
How does the melting point compare to aluminum and titanium?
Aluminum melts at just 1,221°F (660°C). Titanium sits at 3,034°F (1,668°C). Stainless steel lands squarely between them. This middle-ground thermal resistance is one reason stainless dominates applications where aluminum would soften but titanium’s cost — often 10× higher per pound — can’t be justified.
Key Takeaways for Choosing Stainless Steel Based on Thermal Limits
Grade selection starts with one question: what thermal environment will this part actually face? The melting point of stainless steel — ranging from roughly 2,550°F to 2,790°F across all families — sets the absolute ceiling, but it’s rarely the number that matters in practice. Maximum continuous service temperature is the real constraint, and it sits hundreds of degrees below the solidus.
Here’s a quick decision framework by grade family:
- Austenitic (304, 316, 321, 347): Solidus range of 2,540–2,650°F. Best for sustained high-temperature service up to 1,500–1,650°F, especially stabilized grades like 321 and 347 that resist sensitization during prolonged heat exposure.
- Ferritic (430) and Martensitic (410, 420): Solidus range of 2,600–2,790°F. Adequate for moderate temperatures, but ferritics lose toughness above 750°F and martensitic grades sacrifice hardness beyond their tempering limits.
- Duplex (2205): Solidus near 2,550°F. Exceptional strength, but embrittlement risk above 600°F makes it a poor choice for hot service despite its high melting range.
- Precipitation-Hardened (17-4 PH): Solidus around 2,560°F. Useful to about 1,100°F before aging-induced properties degrade.
Two mistakes show up repeatedly in thermal design. First, assuming a higher melting range equals better high-temperature performance — duplex steel disproves that immediately. Second, ignoring the gap between solidus and liquidus during welding, which creates a mushy zone that traps interdendritic defects if heat input isn’t controlled. The ASME Boiler and Pressure Vessel Code publishes allowable stress values at elevated temperatures for exactly this reason: melting data alone doesn’t protect a design.
Match the grade to the service temperature, not the melting point. That single shift in thinking prevents over-specification, reduces material cost, and — most critically — keeps components operating safely within their true thermal limits.
See also
What Is Food Grade Stainless Steel and Why Does It Matter
Ultimate Guide: Laser Welding Heat-Affected Zone
How to Tell Aluminum from Stainless Steel: Shop Guide
How to Work Out the Weight of Stainless Steel Plates with Examples